Calcium hydrogen sulfite
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
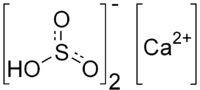
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Calcium hydrogen sulfite | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | Ca (HSO 3 ) 2 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 202.22 g mol −1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Calcium hydrogen sulfite is a chemical compound , more precisely the "acid" calcium salt of sulphurous acid with the chemical formula Ca (HSO 3 ) 2 .
Extraction and presentation
Calcium hydrogen sulfite can be obtained by the action of sulfur dioxide on an aqueous lime solution . It is created (in addition to calcium sulfite ) during flue gas desulfurization .
use
Calcium hydrogen sulfite is used as a food additive . It is used as an antioxidant , preservative and color stabilizer (especially for wines and dried fruits). It is also used in the form of an aqueous solution to dissolve lignin in the production of wood pulp and as a bleaching agent (discoloration of sponges), as an anti-chlorine agent and as a clarifying agent for beverages.
See also
Individual evidence
- ↑ Entry on E 227: Calcium hydrogen sulphite in the European database for food additives, accessed on June 27, 2020.
- ↑ There is not yet a harmonized classification for this substance . A labeling of calcium hydrogen sulphite in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), retrieved on July 28, 2019, is reproduced from a self-classification by the distributor .
- ↑ Technologies to prevent sulfur emissions
- ↑ Charles E.Mortimer and Ulrich Müller: The basic knowledge of chemistry , ISBN 3-13-484308-0 .
