Magnesium nitride
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
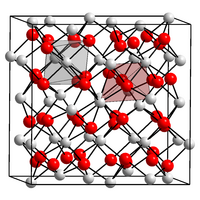
|
||||||||||||||||
| __ N 3− __ Mg 2+ | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Magnesium nitride | |||||||||||||||
| Ratio formula | Mg 3 N 2 | |||||||||||||||
| Brief description |
beige solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 100.95 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
2.71 g cm −3 |
|||||||||||||||
| Melting point |
800 ° C (decomposition) |
|||||||||||||||
| solubility |
reacts with water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Magnesium nitride is a chemical compound made up of the elements magnesium and nitrogen . It has the formula Mg 3 N 2 and belongs to the group of nitrides .
Extraction and presentation
It is formed as a yellow solid, for example when metallic magnesium is heated to approx. 300 ° C under a nitrogen atmosphere.
This reaction is strongly exothermic with −462.8 kJ mol −1 from the solid phase and with −479.6 kJ mol −1 from the liquid phase.
It can also be produced by reacting magnesium and ammonia .
When magnesium metal is burned in air, it is formed alongside magnesium oxide (MgO) and makes it appear yellowish.
properties
Magnesium nitride is a greenish-yellow to yellow-orange colored, loose powder. It has a cubic crystal structure of the carbon sesquioxide type (a = 9.95 Å ). Magnesium nitride hydrolyzes with water to form magnesium hydroxide and ammonia .
In the historical Serpek process , this reaction (in addition to the hydrolysis of AlN) was used to synthesize ammonia (see Haber-Bosch process ).
Individual evidence
- ↑ a b c d data sheet magnesium nitride from AlfaAesar, accessed on December 15, 2010 ( PDF )(JavaScript required) .
- ↑ a b Entry on magnesium nitride in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ↑ a b c Georg Brauer (ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 911.
- ↑ Yuan Chunmiao, Yu Lifu, Li Chang, Li Gang, Zhong Shengjun: Thermal analysis of magnesium reactions with nitrogen / oxygen gas mixtures in J. Hazard. Mat. 260 (2013) 707-714, doi : 10.1016 / j.jhazmat.2013.06.047 .




