Manganese (III) oxide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
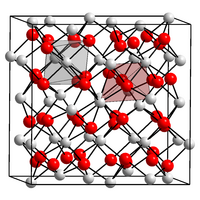
|
||||||||||||||||
| __ Mn 3+ __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Manganese (III) oxide | |||||||||||||||
| other names |
|
|||||||||||||||
| Ratio formula | Mn 2 O 3 | |||||||||||||||
| Brief description |
black odorless solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 157.88 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
4.50 g cm −3 |
|||||||||||||||
| Melting point |
940 ° C (decomposition) |
|||||||||||||||
| solubility |
almost insoluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| MAK |
0.5 mg m −3 |
|||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Manganese (III) oxide is an oxide of manganese . Manganese (III) oxide is known in nature as the rare mineral bixbyite .
Extraction and presentation
It is produced, for example, in zinc-manganese dioxide cells when they are discharged:
Manganese (III) oxide is also formed during the decomposition of manganese (IV) oxide (MnO 2 at temperatures above 535 ° C).
When a solution of manganese sulfate tetrahydrate is reacted with hydrogen peroxide and ammonia at room temperature, γ-MnO (OH) ( manganite ) is obtained. From this, γ-manganese (III) oxide can be obtained by careful dehydration in vacuo at 250 ° C.
properties
Manganese (III) oxide is a non-flammable black odorless solid that is practically insoluble in water. It decomposes when heated above 940 ° C. Γ-Mn 2 O 3 , which does not occur in nature, has a tetragonal crystal structure with the space group I 4 1 / amd (No. 141) (a = 577 pm, c = 944 pm). It converts to the α-form in 48 hours at 500 ° C and in one year at room temperature. At temperatures above 29 ° C, this has a cubic bixbyite structure with space group Ia 3 (no. 206) and below that an orthorhombic structure with space group Pcab (no. 61, position 2) . Other modifications also exist at higher pressures. For example, δ-Mn 2 O 3 , which can be synthesized at pressures above 28 GPa, has a crystal structure of the CaIrO 3 type with the space group Cmcm (No. 63) , ε-Mn 2 O 3 has a crystal structure of the corundum type with the space group R 3 ( No. 148) and ζ-Mn 2 O 3 a distorted triclinic double perovskite structure with the space group F 1 (No. 2, position 5) .
use
Manganese (III) oxide is used as a raw material for the production of lithium manganese (III, IV) oxide LiMn 2 O 4 ( cathode material of lithium-ion batteries ) and as a pigment in dyes (colors glass e.g. brown). The mixed oxide with yttrium and indium results in a brilliant blue pigment, the YInMn blue . The general formula is YIn 1-x Mn x O 3 If it does not contain manganese, the oxide is colorless, if it does not contain indium, it is black.
Individual evidence
- ↑ a b c d e f g h Entry on manganese (III) oxide in the GESTIS substance database of the IFA , accessed on February 1, 2016(JavaScript required) .
- ↑ Chempage: Leclanché cell
- ↑ a b Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1582.
- ↑ Ferrimagnetism in γ-Manganese Sesquioxide (γ - Mn 2 O 3 ) Nanoparticles , Journal of the Korean Physical Society, Vol. 46, No. 4, April 2005, pp. 941-944.
- ↑ S. Geller: Structure of α-Mn2O3, (Mn0.983Fe0.017) 2O3 and (Mn0.37Fe0.63) 2O3 and relation to magnetic ordering. In: Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry. 27, pp. 821-828, doi : 10.1107 / S0567740871002966 .
- ↑ Structure and magnetic properties of Mn2O3 ( Memento of the original from March 4, 2016 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice. (PDF; 713 kB), 2009.09.30 Lab seminar, Kaist, Jo Euna
- ↑ The numbering of this axis position does not correspond to the order of the International Tables for Crystallography , because it is not listed there.
- ↑ Source: A path to new manganites with perovskite structure , Sergey V. Ovsyannikov, Artem M. Abakumov, Alexander A. Tsirlin, Walter Schnelle, Ricardo Egoavil, J. o. Verbeeck, Gustaaf Van Tendeloo, Konstantin V. Glazyrin, Michael Hanfland , Leonid Dubrovinsky: Perovskite-like Mn2O3: A Path to New Manganites. In: Angewandte Chemie International Edition. 52, 2013, pp. 1494–1498, doi : 10.1002 / anie.201208553 .
- ↑ Andrew E. Smith, Hiroshi Mizoguchi, Kris Delaney, Nicola A. Spaldin, Arthur W. Sleight, MA Subramanian: Mn in Trigonal Bipyramidal Coordination: A New Blue Chromophore. In: Journal of the American Chemical Society . 131, 2009, pp. 17084-17086, doi: 10.1021 / ja9080666 .



