Kidney stone reaction
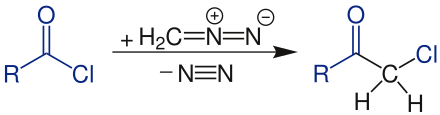
The kidney stone reaction is a name reaction from the field of organic chemistry . It is named after Maximilian Kidney Stone . Starting from a carboxylic acid chloride , an α-chloromethyl ketone is synthesized with diazomethane :
Reaction mechanism
First, the carboxylic acid chloride is reacted with the diazomethane . In the intermediate product formed, the chloride ion is split off by rearrangement , which in the next step attacks the adjacent carbon atom of the diazo group. Nitrogen (N 2 ) is split off and the end product, chloromethyl ketone, is formed.
literature
- Zerong Wang: Comprehensive Organic Name Ractions and Reagents . Vol. 3, p. 2058.
Individual evidence
- ↑ Douglas Arthur Clibbens, Maximilian Kidney Stone: The action of diazomethane on some aromatic acyl chlorides . In: Journal of the Chemical Society, Transactions . tape 107 , no. 0 , 1915, p. 1491-1494 , doi : 10.1039 / CT9150701491 .
- ^ WE Bachmann, WS Struve: The Arndt-Eistert Synthesis . In: Organic Reactions . John Wiley & Sons, Inc., 2004, ISBN 0-471-26418-0 .
- ↑ Yuji Miyahara: Facile synthesis of 2,5-diacylthiophenes . In: Journal of Heterocyclic Chemistry . tape 16 , no. 6 , September 1979, pp. 1147-1151 , doi : 10.1002 / jhet.5570160612 .
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals . 14th Edition, Merck & Co., Whitehouse Station, NJ 2006, ISBN 978-0-911910-00-1 , pp. ONR-66.