Ice VI
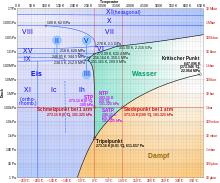
Ice VI is a form of ice that can exist at high pressure on the order of 1 GPa (= 10,000 bar ) and temperatures of approx. 130 to 355 Kelvin (-143 ° C to 82 ° C); see also the phase diagram of water. His discovery and the other high pressure forms of ice were published by PW Bridgman in January 1912.
properties
Eis VI has a density of 1.31 g / cm³ and has a tetragonal crystal system with the space group P4 2 / nmc ; the unit cell contains 10 water molecules and has the dimensions a = 6.27 Å , c = 5.79 Å. With ice VII and liquid water it has a triple point at approx. 82 ° C and 2.22 GPa and with liquid water and ice V at 0.16 ° C and 0.6324 GPa = 6324 bar.
Web links
- Physics of ice (iktp.tu-dresden.de)
- Ice phases (www.idc-online.com)
- Ice VI structure (www1.lsbu.ac.uk)
Individual evidence
- ^ Water, in the Liquid and Five Solid Forms, under Pressure PW Bridgman (1912), www.jstor.org, accessed October 3, 2019
- ↑ Detailed crystallographic analysis of the ice VI to ice XV hydrogen ordering phase transition CG Salzmann (2016), aip.scitation.org, full text on arxiv.org
- ↑ Reports: Structure of Ice VI science.sciencemag.org, B. Kamb, October 8, 1965.
- ↑ Water Phase Diagram www1.lsbu.ac.uk, version of September 9, 2019, accessed on October 3, 2019