Clobenpropit: Difference between revisions
Content deleted Content added
No edit summary Tags: Mobile edit Mobile web edit |
Entranced98 (talk | contribs) +sd |
||
| (9 intermediate revisions by 8 users not shown) | |||
| Line 1: | Line 1: | ||
{{short description|Chemical compound}} |
|||
{{Drugbox |
{{Drugbox |
||
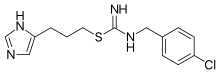
| IUPAC_name = ''N'''-[(4-chlorophenyl)methyl]-1-[3-(3''H''-imidazol-4-yl)propylsulfanyl]formamidine |
| IUPAC_name = ''N'''-[(4-chlorophenyl)methyl]-1-[3-(3''H''-imidazol-4-yl)propylsulfanyl]formamidine |
||
| Line 6: | Line 7: | ||
| tradename = |
| tradename = |
||
| pregnancy_category = |
| pregnancy_category = |
||
| legal_US = IND |
|||
| legal_status = Investigative new drug (not yet approved by the FDA) |
|||
| routes_of_administration = |
| routes_of_administration = |
||
| Line 17: | Line 18: | ||
<!--Identifiers--> |
<!--Identifiers--> |
||
| CAS_number = 145231-45-4 |
| CAS_number = 145231-45-4 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| ⚫ | |||
| UNII = RKU631JF4H |
|||
| ⚫ | |||
| ATC_suffix = |
| ATC_suffix = |
||
| PubChem = 2790 |
| PubChem = 2790 |
||
| Line 26: | Line 29: | ||
<!--Chemical data--> |
<!--Chemical data--> |
||
| C=14 | H=17 | Cl=1 | N=4 | S=1 |
| C=14 | H=17 | Cl=1 | N=4 | S=1 |
||
| molecular_weight = 308.83 g/mol |
|||
| smiles = C1=CC(=CC=C1CN=C(N)SCCCC2=CN=CN2)Cl |
| smiles = C1=CC(=CC=C1CN=C(N)SCCCC2=CN=CN2)Cl |
||
}} |
}} |
||
'''Clobenpropit''' is a [[histamine]] [[H3 antagonist|H<sub>3</sub> receptor |
'''Clobenpropit''' is a [[histamine]] [[H3 antagonist|H<sub>3</sub> receptor antagonist]].<ref name="pmid17537431">{{cite journal | vauthors = Sahlholm K, Nilsson J, Marcellino D, Fuxe K, Arhem P | title = The human histamine H3 receptor couples to GIRK channels in ''Xenopus'' oocytes | journal = European Journal of Pharmacology | volume = 567 | issue = 3 | pages = 206–210 | year = 2007 | pmid = 17537431 | doi = 10.1016/j.ejphar.2007.04.032 }}</ref> It has [[neuroprotective]] effects via stimulation of [[GABA]] release in brain cells ''[[in vitro]]''.<ref>{{ cite journal | vauthors = Dai H, Fu Q, Shen Y, Hu W, Zhang Z, Timmerman H, Leurs R, Chen Z | title = The histamine H3 receptor antagonist clobenpropit enhances GABA release to protect against NMDA-induced excitotoxicity through the cAMP/protein kinase A pathway in cultured cortical neurons | journal = European Journal of Pharmacology | year = 2007 | volume = 563 | issue = 1–3 | pages = 117–123 | pmid = 17350613 | doi = 10.1016/j.ejphar.2007.01.069 }}</ref> |
||
== See also == |
|||
* [[H3 antagonist|H<sub>3</sub> receptor antagonist]] |
|||
== References == |
== References == |
||
{{Reflist}} |
{{Reflist}} |
||
{{Histaminergics}} |
{{Histaminergics}} |
||
| Line 46: | Line 44: | ||
{{ |
{{nervous-system-drug-stub}} |
||
Latest revision as of 01:31, 13 November 2022
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C14H17ClN4S |
| Molar mass | 308.83 g·mol−1 |
| 3D model (JSmol) | |
| |
Clobenpropit is a histamine H3 receptor antagonist.[1] It has neuroprotective effects via stimulation of GABA release in brain cells in vitro.[2]
References[edit]
- ^ Sahlholm K, Nilsson J, Marcellino D, Fuxe K, Arhem P (2007). "The human histamine H3 receptor couples to GIRK channels in Xenopus oocytes". European Journal of Pharmacology. 567 (3): 206–210. doi:10.1016/j.ejphar.2007.04.032. PMID 17537431.
- ^ Dai H, Fu Q, Shen Y, Hu W, Zhang Z, Timmerman H, Leurs R, Chen Z (2007). "The histamine H3 receptor antagonist clobenpropit enhances GABA release to protect against NMDA-induced excitotoxicity through the cAMP/protein kinase A pathway in cultured cortical neurons". European Journal of Pharmacology. 563 (1–3): 117–123. doi:10.1016/j.ejphar.2007.01.069. PMID 17350613.
