Corey-Schmidt oxidation
The Corey-Schmidt oxidation (also PDC oxidation ), named after its discoverers Elias J. Corey and Greg Schmidt, is a name reaction from organic chemistry and was first published in 1979. The reaction describes the oxidation of an alcohol to an aldehyde (or a carboxylic acid ester ). As oxidant which is Cornforth reagent , better known as pyridinium (short PDC) used:
The Corey-Schmidt oxidation is similar to the Corey-Suggs oxidation (PCC oxidation), in which pyridinium chlorochromate is used as the oxidizing agent .
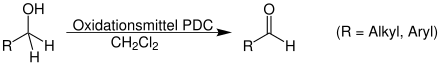
Overview reaction
A primary alcohol RCH 2 OH oxidizes to an aldehyde using dichloromethane and the oxidizing agent pyridinium dichromate:
Instead of the aldehyde, the methyl ester of a carboxylic acid is synthesized when methanol and dimethylformamide are used as solvents:
The oxidation of secondary alcohols R 2 CHOH results in ketones .
Reaction mechanism
The mechanism is described in the literature and is illustrated using the example of an aldehyde synthesis:
A free electron pair of the primary alcohol 1 attacks the chromium atom of the pyridinium dichromate (oxidizing agent) 2 , thereby forming the reactive intermediate 3 . Subsequent electron and atom rearrangement forms intermediate 5 with elimination of a chromic acid derivative 4 . The final cleavage of hydrated chromium (IV) oxide yields the aldehyde 6 .
Individual evidence
- ↑ M. Nakamura, J. Inoue, T. Yamada: A two-step, one-pot synthesis of diverse N-pyruvoyl amino acid derivatives using the Ugi reaction. In: Bioorganic & Medicinal Chemistry Letters , 10 (24), 2000, pp. 2807-2810, doi: 10.1016 / S0960-894X (00) 00577-1 .
- ^ A b E. J. Corey and Greg Schmidt: Useful procedures for the oxidation of alcohols involving pyridinium dichromate in aprotic media. In: Tetrahedron Letters , 20 (5), 1979, pp. 399-402, doi: 10.1016 / S0040-4039 (01) 93515-4 .
- ↑ a b Z. Wang: Comprehensive organic name reactions and reagents Volume 1 . John Wiley, Hoboken (NJ) 2009, ISBN 978-0-470-28662-3 , pp. 738-741 .