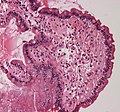
Amyloidosis
| Amyloidosis | |
|---|---|
| Specialty | Endocrinology, cardiology, hematology |
In medicine, amyloidosis refers to a variety of conditions in which amyloid proteins are abnormally deposited in organs and/or tissues, causing disease. A protein is amyloid if, due to an alteration in its secondary structure, it takes on a particular insoluble form, called the beta-pleated sheet.[1]
The term "amyloidosis" refers to a histological finding occurring in several different disease processes that have little in common with each other, and without additional information, the finding is of limited clinical use.[2]
Diagnosis
Amyloid can be diagnosed on microscopic examination of affected tissue. Amyloid deposits can be identified histologically by Congo red staining[3] and viewing under polarized light where amyloid deposits produce a distinctive 'apple green birefringence'. Further, specific tests are available to more precisely identify the amyloid protein. Biopsies are taken from affected organs (for example, the kidney), or often in the case of systemic amyloid, from the rectum, gingiva, or omentum (anterior abdominal adipose tissue). [4]
In addition, all amyloid deposits contain serum amyloid P component (SAP),[5] a circulating protein of the pentraxin family. Radionuclide SAP scans have been developed which can anatomically localize amyloid deposits in patients.
Bleeding under the skin, called amyloid purpura, is seen in a minority of patients with amyloidosis.[6]
Classification of amyloid
Approximately 25 different proteins are known that can form amyloid in humans. Most of them are constituents of the plasma.
Different amyloidoses can be systemic (affecting many different organ systems) or organ-specific. Some are inherited, due to mutations in the precursor protein. Other forms are due to different diseases causing overabundant or abnormal protein production-such as with over production of immunoglobulin light chains in multiple myeloma (termed AL amyloid), or with continuous overproduction of acute phase proteins in chronic inflammation (which can lead to AA amyloid).
There are at least 15 biologically distinct forms of amyloid, some more clinically significant than others. Following is a brief description of the more common types of amyloid:
| Abb. | Amyloid type | Description |
|---|---|---|
| AL | amyloid light chain | Contains immunoglobulin light-chains (λ,κ) derived from plasma cells |
| AA | amyloid associated | Non-immunoglobulin protein made in the liver |
| Aβ | β amyloid | Found in Alzheimer disease brain lesions |
| ATTR | Transthyretin | A mutant form of a normal serum protein that is deposited in the genetically determined familial amyloid polyneuropathies. TTR is also deposited in the heart in senile systemic amyloidosis.[7] |
| Aβ2 m | β2 microglobulin | Not to be confused with Aβ, β2m is a normal serum protein, part of major histocompatability complex (MHC) Class 1 molecules. Can occur in long term haemodialysis. |
| PrP | Prion related protein | In Prion diseases, misfolded prion proteins deposit in tissues and resemble amyloid proteins. |
OMIM includes the following:
| OMIM | Gene | Name | Number |
| Template:OMIM2 | TTR | Senile systemic amyloidosis | (type 1) |
| Template:OMIM2 | GSN | Finnish type amyloidosis | (type 5) |
| Template:OMIM2 | CST3 | Cerebral amyloid angiopathy, Icelandic type | (type 6) |
| Template:OMIM2 | TTR | Leptomeningeal amyloidosis | (type 7) |
| Template:OMIM2 | APOA1, FGA, LYZ | Familial visceral amyloidosis | (type 8) |
| Template:OMIM2 | OSMR | Primary cutaneous amyloidosis | (type 9) |
| Template:OMIM2 | ITM2B | Cerebral amyloid angiopathy, British type | - |
| Template:OMIM2, Template:OMIM2 | APP | Dutch type / Italian type / Iowa type | - |
Classification of amyloidosis
The amyloidoses can be classified as systemic or localised, primary or secondary, or according to which type of amyloid is deposited. By convention, a combination of these approaches are used clinically.
- Systemic amyloidoses are those which affect more than one body organ or system. Localised amyloidoses affect only one body organ or tissue type.
- Primary amyloidoses arise from a disease with disordered immune cell function such as multiple myeloma and other immunocyte dyscrasias.
Secondary (reactive) amyloidoses are those occurring as a complication of some other chronic inflammatory or tissue destructive disease.
- The different types of amyloid deposited (see table, above) may be either primary or secondary, systemic or localised.
Systemic amyloidosis
The common systemic amyloidoses:
| Category | Amyloid type | Precursor protein | Amyloidosis |
|---|---|---|---|
| Systemic acquired | AL | Immunoglobin light chains (Bence Jones protein) | AL amyloidosis (primary amyloidosis) |
| Systemic hereditary | AA | SAA | AA amyloidosis (secondary amyloidosis) |
| Systemic hereditary | Aβ2 m | β2 microglobulin | Haemodialysis associated |
| Systemic hereditary | AA | SAA | Familial mediterranean fever |
| Systemic hereditary | ATTR | transthyretin | Familial amyloidotic polyneuropathies |
| Systemic hereditary | ATTR | transthyretin | Systemic senile amyloidosis |
Organ-specific amyloidosis
In almost all of the organ-specific pathologies, there is significant debate as to whether the amyloid plaques are the causal agent of the disease or instead a downstream consequence of a common idiopathic agent.
Famous People who have contracted Amyloidosis
- Robert Jordan, author of the The Wheel of Time series
- David Lange, Prime Minister of New Zealand
- Richard Caliguiri, mayor of Pittsburgh, Pennsylvania
- Louis J. Tullio, mayor of Erie, Pennsylvania
- Robert P. Casey, Governor of Pennsylvania
- Ed Guthman, Pulitzer Prize winner
Additional images
-
Amyloidosis, dystrophic calcification
-
Small bowel duodenum with amyloid deposition 20X
-
Amyloidosis, Node, Congo Red
-
Amyloidosis, blood vessels, H&E
-
Amyloidosis, lymph node, H&E
-
Amyloidosis, lymph node, polarizer
References
- ^ "Atlas of Pathology".
- ^ Gertz MA (2004). "The classification and typing of amyloid deposits". Am. J. Clin. Pathol. 121 (6): 787–9. doi:10.1309/TR4L-GLVR-JKAM-V5QT. PMID 15198347.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Satoskar AA, Burdge K, Cowden DJ, Nadasdy GM, Hebert LA, Nadasdy T (2007). "Typing of amyloidosis in renal biopsies: diagnostic pitfalls". Arch. Pathol. Lab. Med. 131 (6): 917–22. PMID 17550319.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson; & Mitchell, Richard N. (2007). Robbins Basic Pathology (8th ed.). Saunders Elsevier. p.171 ISBN 978-1-4160-2973-1
- ^ Togashi S, Lim SK, Kawano H; et al. (1997). "Serum amyloid P component enhances induction of murine amyloidosis". Lab. Invest. 77 (5): 525–31. PMID 9389795.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Eder L, Bitterman H (2007). "Image in clinical medicine. Amyloid purpura". N. Engl. J. Med. 356 (23): 2406. doi:10.1056/NEJMicm061510. PMID 17554122.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Hassan W, Al-Sergani H, Mourad W, Tabbaa R (2005). "Amyloid heart disease. New frontiers and insights in pathophysiology, diagnosis, and management". Tex Heart Inst J. 32 (2): 178–84. PMID 16107109.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
External links
- Overview at Mayo Clinic
- Overview at Cleveland Clinic
- [1] Boston University Amyloid Treatment and Research Program
- www.amyloidosisaustralia.org Information, support and advice to anyone with Amyloidosis