G proteins
The term G-protein stands for guanosine triphosphate-binding protein or GTP-binding protein . G proteins occupy a key position in signal transmission ( signal transduction ) between receptor and second messenger systems. A distinction is made between membrane-bound heterotrimeric G-proteins and cytosolic so-called small G-proteins .
Heterotrimeric G proteins
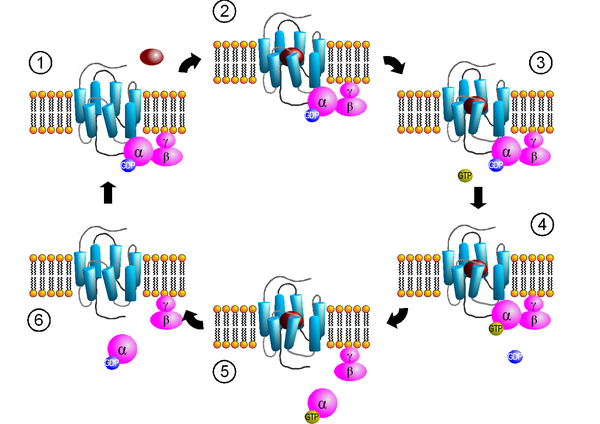
Heterotrimeric G proteins are made up of three subunits (α, β and γ), with the α subunit having a GDP / GTP binding domain. In the inactive form, the α subunit has bound GDP and is associated with βγ subunits. A heterotrimeric G protein can be activated by a G protein-coupled receptor that is or binds to a GTP exchange factor (guanine-nucleotide exchange factor, GEF). The α subunit exchanges its bound GDP for GTP (under the action of the GEF), whereupon the α subunit dissociates from the βγ subunits. The subunits βγ do not disintegrate further, but represent a functional unit. The two released G protein subunits α and βγ can regulate different effectors . Examples are adenylyl cyclases , G-protein-coupled receptor kinases (GRKs) and phospholipases . There are several isoforms of the individual subunits . So far, 21 different α G protein subunits, 5 β subunits and 12 γ subunits are known.
Heterotrimeric G proteins occupy a key position in signal transduction and are responsible for physiological (e.g. sight, smell, blood pressure regulation, etc.) and pathophysiological effects (e.g. arterial hypertension , heart failure ).
Subfamilies
- Gs proteins: cAMP-dependent reactions
- Gi proteins: inhibitory G proteins
- Gq proteins: phospholipase C-coupled G proteins
Small G proteins (monomeric)
Small G-proteins (also small GTP-ases ) are monomeric GTP -binding proteins with a molecular mass of 20 to 40 kDa . Over 100 different small G proteins are currently known, which are divided into 5 families based on phylogenetic similarities and differences: Ras , Rho , Rab, Sar1 / Arf and Ran. They are involved in the regulation of numerous cell functions in the cell cycle , e.g. B. the regulation of gene expression (Ras and Rho), the regulation of the cytoskeleton (Rho), the regulation of vesicle transport (Rab and Sar1 / Arf) as well as the regulation of the transport between the cytoplasm and the nucleus (Ran). The small GTPases change from an inactive, GDP-bound form in the cytosol to an active, GTP-bound form on the plasma membrane. They are inhibited by 14-3-3 proteins .
literature
- K. Aktories, JT Barbieri: Bacterial cytotoxins: targeting eukaryotic switches. In: Nat. Rev. Microbiol. 3, 2005, pp. 397-410.
- K. Burridge, K. Wennerberg: Rho and Rac take center stage. In: Cell . 116, 2004, pp. 167-179.
- HE Hamm: The Many Faces of G Protein Signaling. In: J. Biol. Chem. 273, 2, 1998, pp. 669-672.
- A. Ivetic, AJ Ridley: Ezrin / radixin / moesin proteins and Rho GTPase signaling in leukocytes. In: Immunology. 112, 2004, pp. 165-176.
- LM Machesky, A. Hall: Rho: a connection between membrane receptor signaling and the cytoskeleton. In: Trends Cell Biol . 6, 1996, pp. 304-310.
- HF Paterson, AJ Self, MD Garrett, I. Just, K. Aktories, A. Hall: Microinjection of recombinant p21rho induces rapid changes in cell morphology. In: J. Cell Biol. 111, 1990, pp. 1001-1007.
- M. Raftopoulou, A. Hall: Cell migration: Rho GTPases lead the way. In: Dev. Biol. 265, 2004, pp. 23-32.
- Y. Takai, T. Sasaki, T. Matozaki: Small GTP-binding proteins. In: In: Physiol. Rev. 81, 1, 2001, pp. 153-208.
- I. Vetter, A. Wittinghofer: The guanine nucleotide-binding switch in three dimensions. In: Science . 294, 2001, pp. 1299-1304.
Individual evidence
- ↑ Philippe Riou, Svend Kjaer et al: 14-3-3 Proteins Interact with a Hybrid Prenyl-Phosphorylation Motif to Inhibit G Proteins. In: Cell. 153, 2013, pp. 640-653, doi: 10.1016 / j.cell.2013.03.044 .
Web links
- Jennifer McDowall, Interpro: Protein Of The Month: G-Proteins. (engl.)