Iodoalkanes

Iodomethane , the simplest representative of the iodoalkanes
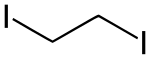
1,2-diiodoethane , with two iodine atoms
The iodoalkanes form a group of halogenated alkanes in which one or more hydrogen atoms have been replaced by iodine atoms . The basic alkane structure can be either straight or branched.
The iodoalkanes can also be divided into primary , secondary and tertiary iodoalkanes.
Manufacturing
The addition of hydrogen iodide to alkenes or 1,3-dienes also leads to iodoalkanes. The electrophilic addition of iodine to alkenes yields diiodalkanes.
Individual evidence
- ↑ Eberhard Breitmaier, Günther Jung: Organic chemistry . 7th edition, Thieme Verlag, 2012, ISBN 978-3-13-541507-9 , p. 185.
- ↑ Eberhard Breitmaier, Günther Jung: Organic chemistry . 7th edition, Thieme Verlag, 2012, ISBN 978-3-13-541507-9 , pp. 186-188.
Web links
Commons : Iodalkane - Collection of images, videos and audio files

