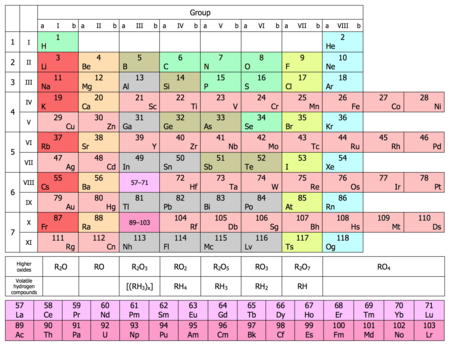
Short period system

The short periodic table is a representation of the periodic table of the elements . In the short period system, main and subgroups are shown nested within one another. When going to press, there was a slight space saving in terms of width.
Above all, there is the possibility of being able to compare the main group and the subgroup. Since the copper group and the zinc group form the 1st and 2nd subgroups in this illustration, the transition from basic to acidic elements is more clearly emphasized than in the long- period system by a diagonal of transition metals running from top left to bottom right . The 8th to 10th subgroups were combined here to form the 8th subgroup, the iron-platinum group ; they are on the far right outside this diagonal.
This representation of the periodic table was mainly used in older textbooks, but can hardly be found today.
literature
- Karlheinz Martin among others: Mathematics, Physics and Chemistry - Grades 7–10. People and Knowledge Volkseigener Verlag Berlin, 1983.
- Karl-Heinz Lautenschläger, Werner Schröter: Pocket book of chemistry. Verlag Harry Deutsch, 2007, ISBN 978-3-8171-1761-1 , p. 88 ff.