Iodanilines
| Iodanilines | ||||||
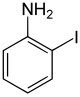
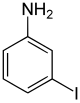
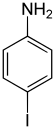
| Surname | 2-iodaniline | 3-iodoaniline | 4-iodoaniline | |||
| other names | o -iodaniline | m -iodaniline | p -iodaniline | |||
| Structural formula |
|
|
|
|||
| CAS number | 615-43-0 | 626-01-7 | 540-37-4 | |||
| PubChem | 11995 | 12271 | 10893 | |||
| Molecular formula | C 6 H 6 IN | |||||
| Molar mass | 219.03 g mol −1 | |||||
| Physical state | firmly | |||||
| Melting point | 55-58 ° C | 21-24 ° C | 61-63 ° C | |||
| boiling point | 145-146 ° C (15 torr ) |
|||||
|
pK s value (of the conjugate acid BH + ) |
2.66 | 3.61 | 3.78 | |||
|
GHS labeling |
|
|
|
|||
| H and P phrases | 302-312-315-332-335 | 302-312-315-332-335 | 302-315-332-335 | |||
| no EUH phrases | no EUH phrases | no EUH phrases | ||||
| 261-280 | 261-280 | 261-305 + 351 + 338 | ||||
The iodoanilines form a group of substances in chemistry that are derived from both aniline and iodobenzene . The structure consists of a benzene ring with attached amino group (-NH 2 ) and iodine (-I) as substituents . Their different arrangement ( ortho , meta or para ) results in three constitutional isomers with the empirical formula C 6 H 6 IN.
Individual evidence
- ↑ a b data sheet 2-iodoaniline from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b data sheet 3-iodoaniline from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b Data sheet 4-iodoaniline from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
