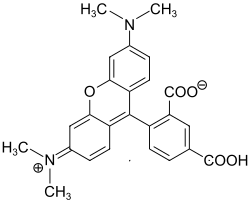
5-carboxytetramethylrhodamine
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||
| General | |||||||||||||
| Surname | 5-carboxytetramethylrhodamine | ||||||||||||
| other names |
5-TAMRA |
||||||||||||
| Molecular formula | C 25 H 22 N 2 O 5 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 430.45 g mol −1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
5-carboxytetramethylrhodamine ( 5-TAMRA ) is a red fluorescent dye from the group of rhodamines . It can be classified both in the group of triphenylmethane dyes and xanthene dyes . 5 ‑ TAMRA is used for fluorescent labeling of nucleic acids and proteins . The carboxy group can be converted into a reactive intermediate state with coupling reagents . Alternatively, the succinimidyl ester 5-carboxytetramethylrhodamine- N- succinimidyl ester ( 5-TAMRA-SE) can be used for marking. 5-TAMRA is used, among other things, for fluorescent substrates of enzymes , hybridization probes and FRET .
Individual evidence
- ↑ a b c Data sheet 5-Carboxytetramethylrhodamine from Sigma-Aldrich , accessed on October 31, 2016 ( PDF ).
- ↑ Maksim V. Kvach, Dmitry A. Tsybulsky, Vadim V. Shmanai, Igor A. Prokhorenko, Irina A. Stepanova, Vladimir A. Korshun: Current Protocols in Nucleic Acid Chemistry . John Wiley & Sons, Inc., 2001, ISBN 978-0-471-14270-6 , chapter Non-Nucleoside Phosphoramidites of Xanthene Dyes (FAM, JOE, and TAMRA) for Oligonucleotide Labeling , doi : 10.1002 / 0471142700.nc0455s52 , PMID 23512693 .
- ↑ GH Lee, EJ Lee, SS Hah: TAMRA- and Cy5-labeled probe for efficient kinetic characterization of caspase-3. In: Analytical biochemistry . Volume 446, February 2014, pp. 22-24; doi: 10.1016 / j.ab.2013.10.013 , PMID 24144487 .