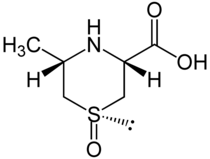
Cycloalline
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||
| General | ||||||||||
| Surname | Cycloalline | |||||||||
| other names |
(1 S , 3 R , 5 S ) -5-methyl-1,4-thiazane-3-carboxylic acid 1-oxide |
|||||||||
| Molecular formula | C 6 H 11 NO 3 S | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 177.23 g mol −1 | |||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Cycloalliin is a heterocyclic compound that, like alliin, is a natural ingredient in various types of leek . It was first isolated from garlic in 1959 ; this contains an average of 0.1% cycloalline. In contrast to alliin, cycloalliin does not react with the alliin-cleaving enzyme alliinase (alliin lyase).
Derived connections
- Cycloalliin- hydrochloride mono hydrate
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ KJ Palmer and KS Lee: The structure of cycloalliin hydrochloride monohydrate , Acta Cryst. (1966). 20, 790-795 doi : 10.1107 / S0365110X66001865
- ↑ KJ Palmer and KS Lee: Erratum: the structure of cycloalliin hydrochloride monohydrate , Acta Cryst. (1972). B28, 2615 doi : 10.1107 / S0567740872006557