Iodonitrobenzenes
| Iodonitrobenzenes | |||||||
| Surname | 2-iodonitrobenzene | 3-iodonitrobenzene | 4-iodonitrobenzene | ||||
| other names | o -iodonitrobenzene | m -iodonitrobenzene | p -iodonitrobenzene | ||||
| Structural formula |
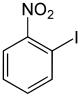
|

|
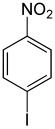
|
||||
| CAS number | 609-73-4 | 645-00-1 | 636-98-6 | ||||
| PubChem | 69115 | 12574 | 12495 | ||||
| Molecular formula | C 6 H 4 INO 2 | ||||||
| Molar mass | 249.01 g mol −1 | ||||||
| Physical state | firmly | ||||||
| Melting point | 49-51 ° C | 35-38 ° C | 171-173 ° C | ||||
| boiling point | 288-289 ° C | 279-280 ° C | 288-290 ° C | ||||
|
GHS labeling |
|
|
|
||||
| H and P phrases | 302-312-315-319-332-335 | 228-302-312-315-319-332-335 | 302-312-315-319-332-335 | ||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||
| 261-280-305 + 351 + 338 | 210-261-280-305 + 351 + 338 | 261-280-305 + 351 + 338 | |||||
The iodonitrobenzenes form a group of substances in chemistry that are derived from both nitrobenzene and iodobenzene . The structure consists of a benzene ring with attached nitro group (–NO 2 ) and iodine (–I) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 6 H 4 INO 2 .
properties
The p -iodonitrobenzene, which has the highest symmetry, has the highest melting point.
Individual evidence
- ↑ a b c d e f Alfa-Aesar catalog 2008/09, p. 1153.
- ↑ Data sheet 1-Iodo-2-nitrobenzene from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ Data sheet 1-Iodo-3-nitrobenzene from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ Data sheet 1-Iodo-4-nitrobenzene from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).

