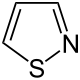
Isothiazole
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Isothiazole | |||||||||||||||
| other names |
1,2-thiazole |
|||||||||||||||
| Molecular formula | C 3 H 3 NS | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 85.13 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| boiling point |
114 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Isothiazole is one of thiophene and pyrrole derived heterocycle . In contrast to the isomeric thiazole , the sulfur atom is next to the nitrogen.
See also
literature
- S. Califano, F. Piacenti, G. Sbrana: The vibrational spectrum of isothiazole. In: Spectrochimica Acta . 20 (3), 1964, pp. 339-344; doi : 10.1016 / 0371-1951 (64) 80031-X .
Individual evidence
- ^ DW Brown and M. Sainsbury: Isothiazoles (PDF; 832 kB), p. 513.
- ↑ There is not yet a harmonized classification for this substance . A labeling of 1,2-thiazole in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), retrieved on July 19, 2019, is reproduced from a self-classification by the distributor .

