Conformation

The conformation of an organic molecule describes the spatial arrangement of its rotatable bonds on the carbon atoms. They completely describe the three-dimensional spatial coordinates of all atoms in the molecule.
The conformation thus also contains information about the stereochemistry , i.e. the configuration , of all stereotopic atoms and about the constitution of the molecule.
Molecules with the same configuration, but which differ in the specific arrangement of the atoms and lie in a minimum energy, are called conformers ; The term rotamer is also used synonymously . The sawhorse and Newman projections can be used to represent them. If there are different conformers in equilibrium with one another, the quantitative ratio is given by a Boltzmann statistic . In contrast to conformers, there is configuration isomerism when the chemical bonds in question cannot be rotated and the isomers can only be converted into one another by breaking the bond.
Conformations in linear molecules
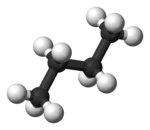
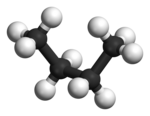
In the example, the staggered conformation of ethane is shown on the left and the ecliptic conformation of the ethane in Newman projection on the right. Underneath there is a representation in the ball-and-stick model .
Depending on the dihedral angle , the conformations of butane are further differentiated into staggered (anti) at a dihedral angle of 180 °, partially hidden at 120 °, skewed (gauche) at 60 ° and ecliptic at a dihedral angle of 0 °.
However, the conformers cannot be isolated, but can only be detected spectroscopically at low temperatures.
Conformations in cyclic molecules

Cycloalkanes can often occur in different conformations that are separated from one another by a significant energy barrier, so that the conformers can be detected next to one another.
The twist , tub, and chair conformations are conformations of cyclic molecules such as cyclohexane . Compared to the tub conformation or the boat conformation , the twist conformation is slightly twisted. As a result, the interactions between the two H atoms, which sit at the two tips of the tub, become weaker and the Pitzer voltage smaller (the H atoms are not exactly eclipsed). The Baeyer tension increases slightly due to the slight twisting of the ring, but this compensates for the van der Waals repulsion between the two H atoms located at the tips of the tub. The twist conformation is therefore about 5.9 kJ / mol (1.4 kcal / mol ) more stable than the tub shape and, like the armchair shape, represents a conformer. Since the tub shape is also more energetic than the armchair conformation, compounds such as cyclohexane occur almost exclusively the almost tension-free chair conformation. Because it is possible to twist the molecule in two directions, there are two energetically equivalent twist conformations.
Ring inversion
The ring inversion (engl. Ring flip ) is a phenomenon in which cyclic by rotation about single bonds conversion conformers same ring shapes takes place. The spatial positions of the substituents do not necessarily have to remain the same.
Cyclohexane is the most prominent compound in which ring inversion can be observed. The preferred conformation of this six-membered ring alkane is the chair conformation. Contrary to expectations, the 1 H-NMR spectrum of cyclohexane does not show two signals at different resonance frequencies for axial and equatorial hydrogen atoms, which are in slightly different chemical environments. The ecliptic transition state that is passed through during the conformational change is energetically slightly increased, so that the isomerization takes place quickly at room temperature and only one signal can be observed. This changes when the temperature is lowered so much that the conversion takes place much more slowly due to the energy barrier of the isomerization. In the ring inversion of cyclohexane, all axial substituents become equatorial and all equatorial become axial.
The ring inversion of cyclohexane begins with the transition from the armchair to the half-chair to the twist conformation, with the maximum energy being in the half-chair conformation. Via the intermediate step of the tub conformation, the twist conformation is converted into the mirror-image twist conformation. The tub conformation represents the energy maximum. The armchair conformation after ring inversion is achieved via another half armchair.
Various substituents prevent ring inversion from occurring, for example a second ring attached as in trans -decalin . Acyclic substituents can also have this effect. The tert -butyl group in tert -butylcyclohexane allows only one conformation; the substituent is therefore also referred to as a conformational anchor. Due to its high steric demand, it causes far greater 1,3-diaxial strain in the axial position than the three hydrogen substituents when it is equatorial. The “underside” does not change with the ring inversion of tert- butylcyclohexane.
In tetrahydropyran , an oxygen atom replaces a carbon atom of the cyclohexane ring, so that the chair conformers are asymmetrical. This can also be illustrated by a special notation. If you think of a plane that is formed by three carbon atoms and one oxygen atom, there is one carbon atom below and one above this plane (see picture). When the ring atoms are numbered, the ring is formed by the atoms C2-C3-C5-O; C1 is below, C4 above that level. This is then referred to as the 4 C 1 conformation. The alternative chair (in the picture on the right) then has the 1 C 4 conformation.

The substituted tetrahydropyran D - glucose preferably adopts the 4 C 1 conformation in which the largest substituent, the hydroxymethyl group, is in the equatorial position (cf. Fig. 1). In the 1 C 4 conformation (2), however, this would be positioned axially, so that in β- D -glucose it interacts with two further substituents (1,3- syn -axial). This results in a large 1,3-diaxial stress. Other hydroxyl groups also interact axially with one another. Overall, these interactions destabilize the 1 C 4 conformation.
In glucopyranoses, the largest substituent is therefore in the equatorial position, if this results in as few steric interactions as possible with other substituents. This is generally the case with D sugars due to the 4 C 1 conformation. In L sugar, on the other hand, the hydroxymethyl group likes to position itself equatorially in the 1 C 4 conformation.
Metallocenes
Metallocenes such as B. Ferrocene can exist in two different conformations.
See also
Individual evidence
- ↑ see chemistry: the basic knowledge of chemistry (Charles E. Mortimer, Ulrich Müller, Thieme (9th edition), page 529).
- ↑ Eberhard wide Maier, Günther Jung: Organic Chemistry , Issue 5, Georg Thieme Verlag, 2005, ISBN 978-3-13-541505-5 , Google Books .
- ↑ Momcilo Miljkovic: Carbohydrates: Synthesis, Mechanisms, and Stereoelectronic Effects . Springer, Berlin 2009; P. 41.
- ↑ Momcilo Miljkovic: Carbohydrates: Synthesis, Mechanisms, and Stereoelectronic Effects . Springer, Berlin 2009; P. 40.
- ^ NN Greenwood and A. Earnshaw: Chemistry of the Elements , 1st Edition, 1988, pp. 408-409, ISBN 3-527-26169-9 .
swell
- Clayden, J. Greeves, N. Warren, S. Wothers, P. (2001). Organic Chemistry , Oxford, ISBN 0-19-850346-6 .