Reaction parameters (chemistry)
Reaction parameters are physical quantities that influence the course of a reaction. This includes:
Pressure , temperature , volume , catalyst or enzyme , influence of light , amount of substance , solvent , partial pressure (for gases), pH value , ultrasound and microwaves, etc.
There are reactions that are carried out under inert gas , which would also be noted under the reaction parameters.
Significance for the chemical equilibrium of a reaction
Reaction parameters play a role for the reaction profile, the reaction rate and the reaction kinetics . According to the Le Chatelier principle, they shift the equilibrium of a chemical reaction. An important criterion for the course of a chemical reaction is the change in the free enthalpy of reaction Δ G, described by the basic equation:
- Δ G = Δ H -T Δ S
(S = entropy)
The free enthalpy is in turn related to the chemical equilibrium.
The following applies:
- Δ G = RT ln K, where K is the equilibrium constant.
Reactions can be endothermic or exothermic, depending on whether they have a positive or negative reaction enthalpy Δ H, which in turn can be influenced by varying the reaction parameters.
For example, catalysts can lower the activation energy of a reaction.
Notation
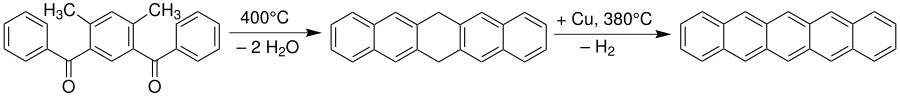
The reaction parameters are noted in a reaction equation above or below the reaction arrow:
Not to be confused with parameters from theoretical chemistry
There are geometric parameters for the reaction coordinate of a chemical reaction in order to calculate energy hypersurfaces.
literature
- D. Schaum, JL Rosenberg: Exercises in General Chemistry , Mc Graw Hill, 1982, ISBN 0-07-084374-0 .