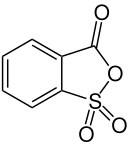
2-sulfobenzoic anhydride
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 2-sulfobenzoic anhydride | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 7 H 4 O 4 S | ||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 184.17 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
116-122 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
2-sulfobenzoic anhydride is the internal anhydride of 2-sulfobenzoic acid . It is used for the synthesis of sulfonphthaleins .
The hydroxyl group content of a sample can be determined analytically by esterification with sulfobenzoic anhydride . Analogously, the content of primary and secondary amino groups of a substance to be examined can be determined analytically by amide formation with sulfobenzoic anhydride .
Individual evidence
- ↑ a b c Data sheet 2-sulfobenzoic anhydride (PDF) from Merck , accessed on September 25, 2016.
- ^ A b V. Iyer, NK Mathur: Determination of purity of o-sulphobenzoic anhydride . In: Talanta . tape 13 , no. 11 , 1966, pp. 1592-1595 , doi : 10.1016 / 0039-9140 (66) 80108-X .
- ↑ Esterification of Hydroxylated Polymers with 2-Sulfobenzoic Acid Cyclic Anhydride , 2005.
