4-aminonaphthalene-1-sulfonic acid
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
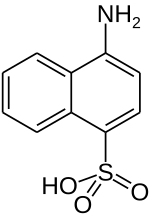
|
||||||||||
| General | ||||||||||
| Surname | 4-aminonaphthalene-1-sulfonic acid | |||||||||
| Molecular formula | C 10 H 9 NO 3 S | |||||||||
| Brief description |
white solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 223.25 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| density |
1.67 g cm −3 |
|||||||||
| Melting point |
> 300 ° C |
|||||||||
| solubility |
very sparingly soluble in water (0.31 g l −1 at 20 ° C) |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
4-aminonaphthalene-1-sulfonic acid is a chemical compound from the group of aminosulfonic acids .
Extraction and presentation
4-aminonaphthalene-1-sulfonic acid can be obtained by reacting 1-aminonaphthalene with sulfuric acid.
properties
4-aminonaphthalene-1-sulfonic acid is a flammable, hardly inflammable white solid that is very sparingly soluble in water.
use
4-aminonaphthalene-1-sulfonic acid is used as an intermediate in the synthesis of azo dyes (such as Rocceline , aka Solid Red A). Its sodium salt has been used as a non-toxic hemostatic agent.
Individual evidence
- ↑ a b c d e f g h Entry on 4-aminonaphthalene-1-sulfonic acid in the GESTIS substance database of the IFA , accessed on December 31, 2016(JavaScript required) .
- ↑ a b Gerald Booth: Ullmann's Encyclopedia of Industrial Chemistry . Wiley-VCH Verlag & Co. KGaA, 2000, ISBN 978-3-527-30673-2 , Naphthalene Derivatives.
- ↑ Data sheet 4-Amino-1-naphthalenesulfonic acid, 97% from Sigma-Aldrich , accessed on December 31, 2016 ( PDF ).
