Borabenzene
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||
| Borabenzene coordinates to pyridine or triphenylphosphine | ||||||||||
| General | ||||||||||
| Surname | Borabenzene | |||||||||
| other names |
Borinine |
|||||||||
| Molecular formula | C 5 H 5 B | |||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 75.90 g mol −1 | |||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Borabenzene is a heteroaromatic compound in which a boron atom replaces a methine (CH) group in a benzene molecule. Free borabenzene - without an electron donor ligand on the boron atom - has not yet been isolated, although the chemical bond between boron and carbon atoms is relatively stable. The instability of borabenzene is the result of the high Lewis acidity of the boron atom due to its electron deficiency. Borabenzene reacts with pyridine to form a stable adduct that is isoelectronic with biphenyl .
properties
Borabenzene as a ligand
Borabenzene acts as a π donor in complexes - similar to a cyclopentadienyl anion . Numerous transition metal complexes of borabenzene are known.
Reactions
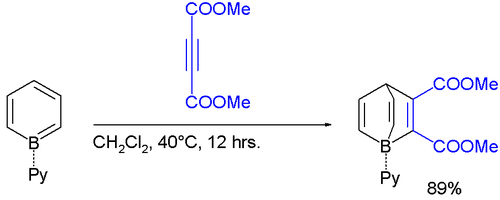
Borabenzene reacts with electron-poor alkynes in a Diels-Alder reaction :
Analog connections
A number of similar heteroaromatic bora compounds are known, inter alia
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Jane E. Macintyre: Dictionary of Organometallic Compounds . CRC Press, 1994, ISBN 978-0-412-43060-2 , pp. 482 ( limited preview in Google Book search).
- ↑ Thomas K. Wood, Warren E. Piers, Brian A. Keay, Masood Parvez: 1-Borabarrelene Derivatives via Diels-Alder Additions to Borabenzenes Org. Lett. ; 2006 ; 8 (13) pp. 2875-2878, doi : 10.1021 / ol061201w .