Diiodobenzenes
| Diiodobenzenes | |||||||
| Surname | 1,2-diiodobenzene | 1,3-diiodobenzene | 1,4-diiodobenzene | ||||
| other names | o -diiodobenzene | m -diiodobenzene | p -diiodobenzene | ||||
| Structural formula |
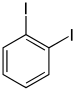
|

|

|
||||
| CAS number | 615-42-9 | 626-00-6 | 624-38-4 | ||||
| PubChem | 11994 | 12270 | 12208 | ||||
| Molecular formula | C 6 H 4 I 2 | ||||||
| Molar mass | 329.91 g mol −1 | ||||||
| Physical state | liquid | firmly | |||||
| Melting point | 27 ° C | 40 ° C | 131-133 ° C | ||||
| boiling point | 287 ° C | 285 ° C | 285 ° C | ||||
| density | 2.524 g cm −3 | ||||||
|
GHS labeling |
|
|
|
||||
| H and P phrases | 315-318-335 | 315-319-335 | no H-phrases | ||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||
| 261-280-305 + 351 + 338 | 261-305 + 351 + 338 | no P-phrases | |||||
In chemistry , the diiodobenzenes form a group of substances consisting of a benzene ring with two iodine atoms (–I) as substituents . Their different arrangement ( ortho , meta or para ) results in three constitutional isomers with the empirical formula C 6 H 4 I 2 .
properties
The boiling points of the three isomers are relatively close to one another, while their melting points differ more clearly. The 1,4-diiodobenzene, which has the highest symmetry, has the highest melting point.
presentation
The diiodobenzenes can be obtained from the corresponding diaminobenzenes by diazotization and subsequent reaction with copper (I) iodide .
Individual evidence
- ^ A b Morrison / Boyd: Textbook of Organic Chemistry , 3rd Edition, VCH, Weinheim 1986, ISBN 3-527-26067-6 , p. 1113.
- ↑ a b CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
- ↑ a b c data sheet 1,4-diiodobenzene from Sigma-Aldrich , accessed on November 7, 2016 ( PDF ).
- ↑ a b data sheet 1,2-diiodobenzene from Sigma-Aldrich , accessed on February 25, 2011 ( PDF ).
- ↑ Data sheet 1,3-diiodobenzene from Sigma-Aldrich , accessed on February 25, 2011 ( PDF ).
literature
- Abil E. Aliev, Kenneth DM Harris, Xavier Alcobéa, Eugènia Estop: "Dynamic properties of p -diiodobenzene investigated by solid-state 2 H and 13 C nuclear magnetic resonance spectroscopy", in: J. Chem. Soc., Faraday Trans. , 1993 , 89 , pp. 3797-3800; doi : 10.1039 / FT9938903797 .
- TV Magdesieva, II Kukhareva, GA Artamkina, IP Beletskaya, KP Butin: “Electrochemically induced aromatic nucleophilic substitution in p -diiodobenzene with [(η 5 -C 5 H 5 ) Fe (CO) 2 ] - and [(η 5 -C 5 H 5 ) W (CO) 3 ] - -anions ”, in: Journal of Organometallic Chemistry , 1995 , 487 (1-2), pp. 163-166; doi : 10.1016 / 0022-328X (94) 05077-O .
- Linda C. Brazdil, Carlo J. Cutler: “Selective Production of Diiodobenzene and Iodobenzene from Benzene”, in: J. Org. Chem. , 1996 , 61 (26), pp. 9621-9622; doi : 10.1021 / jo961493m .

