Hydroxybenzyl alcohols
| Hydroxybenzyl alcohols | |||||||
| Surname | 2-hydroxybenzyl alcohol | 3-hydroxybenzyl alcohol | 4-hydroxybenzyl alcohol | ||||
| other names |
o -hydroxybenzyl alcohol , 2-methylolphenol, salicyl alcohol , saligenin |
m -hydroxybenzyl alcohol, 3-methylolphenol |
p -hydroxybenzyl alcohol, 4-methylol phenol |
||||
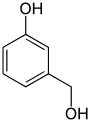
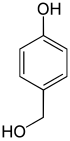
| Structural formula |

|
|
|
||||
| CAS number | 90-01-7 | 620-24-6 | 623-05-2 | ||||
| PubChem | 5146 | 102 | 125 | ||||
| Molecular formula | C 7 H 8 O 2 | ||||||
| Molar mass | 124.14 g mol −1 | ||||||
| Physical state | firmly | ||||||
| Melting point | 83-85 ° C | 69-72 ° C | 114-122 ° C | ||||
| boiling point | |||||||
| pK s value | 9.92 | 9.83 | 9.82 | ||||
|
GHS labeling |
|
|
|
||||
| H and P phrases | 315-319-335 | 315-318-335 | 319 | ||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||
| 261-305 + 351 + 338 | 261-280-305 + 351 + 338 | 305 + 351 + 338 | |||||
The hydroxybenzyl alcohols (less often methylolphenols ) form a group of substances in chemistry that is derived from both benzyl alcohol and phenol . The structure consists of a benzene ring with attached hydroxymethyl (-CH 2 OH) and hydroxyl group (-OH) as substituents . Their different arrangements ( ortho , meta or para ) result in three constitutional isomers with the empirical formula C 7 H 8 O 2 . The 2-hydroxybenzyl alcohol is known under its common name salicyl alcohol . In their acidity they differ little from the phenol (pK s value = 9.99).
Individual evidence
- ↑ a b Data sheet 2-Hydroxybenzyl alcohol from Sigma-Aldrich , accessed on November 3, 2016 ( PDF ).
- ↑ a b Data sheet 3-Hydroxybenzyl alcohol from Sigma-Aldrich , accessed on November 3, 2016 ( PDF ).
- ↑ a b Data sheet 4-Hydroxybenzyl alcohol from Sigma-Aldrich , accessed on November 3, 2016 ( PDF ).
- ↑ a b CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
See also
Web links
Commons : Hydroxybenzyl Alcohols - Collection of pictures, videos, and audio files

