n -propylphenols
| n -propylphenols | |||||||
| Surname | 2- n -propylphenol | 3- n -propylphenol | 4- n -propylphenol | ||||
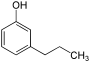
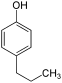
| Structural formula |

|
|
|
||||
| CAS number | 644-35-9 | 621-27-2 | 645-56-7 | ||||
| PubChem | 12570 | 69302 | 12580 | ||||
| Molecular formula | C 9 H 12 O | ||||||
| Molar mass | 136.19 g mol −1 | ||||||
| Physical state | liquid | firmly | |||||
| description | oily liquid | ||||||
| Melting point | 26 ° C | 22 ° C | |||||
| boiling point | 224-226 ° C | 229-231 ° C | 232 ° C | ||||
| density | 0.989 g cm −3 (20 ° C) | 0.95 g cm −3 | 0.983 g cm −3 (25 ° C) | ||||
|
GHS labeling |
|
|
|
||||
| H and P phrases | 302-312-315-319-332-335 | 302-312-314 | 302-312-315-319-332-335 | ||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||
| 261-280-305 + 351 + 338 |
260-303 + 361 + 353-305 + 351 + 338 301 + 330 + 331-405-501 |
261-280-305 + 351 + 338 | |||||
In chemistry, the n- propylphenols form a group of substances that are derived from both phenol and n- propylbenzene . The structure consists of a benzene ring with attached hydroxyl group (-OH) and n -propyl group (-CH 2 -CH 2 -CH 3 ) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 9 H 12 O.
properties
3- n -Propylphenol gives iron (III) chloride in ethanol a green color, in water a bluish color. The methyl ethers, the n- propyl anisoles, are formed by methylation with dimethyl sulfate .
Individual evidence
- ^ A b c Heilbron: Dictionary of Organic Compounds , Volume Four, 1953 , p. 255; Full text .
- ↑ a b Zvi Rappoport (Ed.): CRC Handbook of Tables for Organic Compound Identification . 3 rd Edition, CRC Press / Taylor and Francis, Boca Raton, FL, 1967, ISBN 0-8493-0303-6 , Organic Derivatives of Phenols, S. 96th
- ↑ a b c data sheet 2-propylphenol from Sigma-Aldrich , accessed on August 8, 2017 ( PDF ).
- ↑ a b Data sheet 3-n-Propylphenol from AlfaAesar, accessed on August 8, 2017 ( PDF )(JavaScript required) .
- ↑ a b c data sheet 4-propylphenol from Sigma-Aldrich , accessed on August 8, 2017 ( PDF ).

