Neocuproin
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
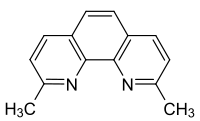
|
||||||||||
| General | ||||||||||
| Surname | Neocuproin | |||||||||
| other names |
2,9-dimethyl-1,10-phenanthroline |
|||||||||
| Molecular formula | C 14 H 12 N 2 | |||||||||
| Brief description |
colorless powder |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 208.26 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| Melting point |
161-163 ° C |
|||||||||
| solubility |
slightly soluble in water |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Neocuproin is a methylated derivative of phenanthroline . It is a chelate complexing agent that can specifically complex copper (I) ions .
Neocuproin is used to mask copper in the titrimetric determination of iron in blood serum with ferrozine as a complexing agent for Fe 2+ . Since this reaction takes place in the micro range, trace elements such as copper (which is also contained in blood serum) would falsify the measurement result.
Web links
- Entry on Neocuproin at TCI Europe, accessed on June 25, 2011.
Individual evidence
- ↑ a b c Data sheet Neocuproin (PDF) from Merck , accessed on April 2, 2010.
- ↑ a b Datasheet Neocuproine from Sigma-Aldrich , accessed on June 16, 2011 ( PDF ).
- ^ Daniel C. Harris: Textbook of Quantitative Analysis , 1998.
