Oxamniquin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
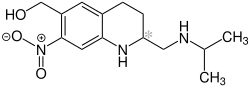
|
|||||||||||||||||||
| Structural formula without stereochemistry | |||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Oxamniquin | ||||||||||||||||||
| Molecular formula | C 14 H 21 N 3 O 3 | ||||||||||||||||||
| Brief description |
orange crystals |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| Drug information | |||||||||||||||||||
| ATC code | |||||||||||||||||||
| Drug class | |||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 279.3 g · mol -1 | ||||||||||||||||||
| Melting point | |||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Oxamniquin is an anthelmintic with schistosomicidal activity against Schistosoma mansoni , but not against other Schistosoma spp. It is a potent single-dose drug used to treat infections in humans. It causes the worms to migrate from the mesenteric veins to the liver, where the male worms remain. The female worms return to the mesentery but are no longer able to lay eggs.
Stereochemistry
Oxamniquine contains a stereocenter and consists of two enantiomers. This is a racemate , i.e. a 1: 1 mixture of ( R ) - and ( S ) -form:
| Enantiomers of Oxamniquine | |
|---|---|
 ( R ) shape |
 ( S ) shape |
Individual evidence
- ↑ a b c Kuhnert-Brandstätter, M .; Völlenklee, R .: Contribution to the polymorphism of medicinal substances, Part 4: Oxamniquin, Resorantel, Spiperon, Suloctidil, Ticlopidinhydrochlorid, Parsol 1789 and Testosteroncyptonat in Sci. Pharm. 55 (1987) 27-39.
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals , 14th Edition (Merck & Co., Inc.), Whitehouse Station, NJ, USA, 2006; Pp. 1192-1193, ISBN 978-0-911910-00-1 .
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Martidale: The Extra Pharmacopoeia , 31st edition, p 121st