Phenyl group
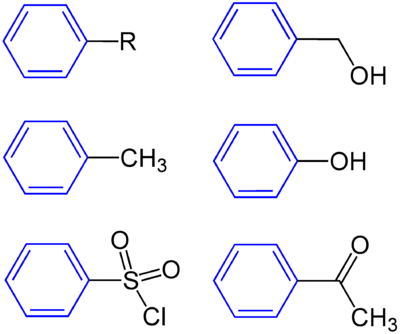
Compounds with a phenyl group ( blue ) with R = organyl group (alkyl group, aryl group, acyl group etc.), halogen, hydroxyl radical, metal etc .; Benzyl alcohol , toluene , phenol , benzenesulfonic acid chloride and acetophenone (from top left to bottom right).
In organic chemistry, the phenyl group is the benzene residue , i.e. the atomic group –C 6 H 5 . In complex chemical formulas , the phenyl group is sometimes abbreviated as Ph , more rarely with Ø or φ. Phenyls have a weak electron-donating effect (see mesomeric effect ).
Examples of compounds with phenyl groups are for example biphenyl , benzoic acid or phenol . The benzyl group (-CH 2 Ph) and the benzoyl group (-C (O) Ph) are extensions of the phenyl group; The phenylene group (–C 6 H 4 -) is also structurally similar .
literature
- Klaus Schwetlick: Organikum . 23rd edition. Wiley-VCH, Weinheim 2009, ISBN 978-3-527-32292-3 .
Individual evidence
- ↑ Otto-Albrecht Neumüller (Ed.): Römpps Chemie-Lexikon. Volume 4: M-Pk. 8th revised and expanded edition. Franckh'sche Verlagshandlung, Stuttgart 1985, ISBN 3-440-04514-5 , p. 3137.