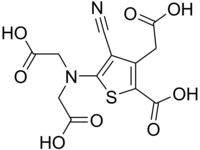
Ranelic acid
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||
| General | |||||||||||||
| Non-proprietary name | Ranelic acid | ||||||||||||
| other names | |||||||||||||
| Molecular formula | C 12 H 10 N 2 O 8 S | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 342.28 g · mol -1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Ranelic acid is the non-proprietary name for the organic chemical compound 5- [bis (carboxymethyl) amino] -2-carboxy-4-cyano-3-thiophene-acetic acid. This is one of the thiophene derivatives, carboxylic acids and nitriles . The salts of ranelic acid are called ranelates.
The strontium salt strontium ranelate C 12 H 6 N 2 O 8 SSr 2 is used as a medicinal substance for the treatment of osteoporosis . The ranelic acid anion (ranelate) serves as a counterion for two strontium cations and brings about favorable bioavailability and compatibility of the pharmacologically active strontium. Ranelic acid itself has no effect on bone metabolism. Ranelic acid is very polar and is hardly absorbed; an accumulation was not observed either in animals or in humans.
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals , 14th Edition (Merck & Co., Inc.), Whitehouse Station, NJ, USA, 2006; P. 1396, ISBN 978-0-911910-00-1 .