Cholesterol 7 alpha-hydroxylase: Difference between revisions
Citation bot (talk | contribs) m Add: url. | You can use this bot yourself. Report bugs here.| Activated by User:Nemo bis | via #UCB_webform |
Rescuing 0 sources and tagging 1 as dead.) #IABot (v2.0.9.5) (Maxim Masiutin - 17931 |
||
| (35 intermediate revisions by 16 users not shown) | |||
| Line 1: | Line 1: | ||
{{Short description|Protein-coding gene in the species Homo sapiens}} |
|||
{{cs1 config|name-list-style=vanc}} |
|||
{{Infobox gene}} |
{{Infobox gene}} |
||
'''Cholesterol 7 alpha-hydroxylase''' also known as '''cholesterol 7-alpha-monooxygenase''' or '''cytochrome P450 7A1''' (CYP7A1) is an [[enzyme]] that in humans is encoded by the |
'''Cholesterol 7 alpha-hydroxylase''' also known as '''cholesterol 7-alpha-monooxygenase''' or '''cytochrome P450 7A1''' (CYP7A1) is an [[enzyme]] that in humans is encoded by the {{gene|CYP7A1}} [[gene]]<ref name="pmid1358792">{{cite journal | vauthors = Cohen JC, Cali JJ, Jelinek DF, Mehrabian M, Sparkes RS, Lusis AJ, Russell DW, Hobbs HH | title = Cloning of the human cholesterol 7 alpha-hydroxylase gene (CYP7) and localization to chromosome 8q11-q12 | journal = Genomics | volume = 14 | issue = 1 | pages = 153–61 | date = Sep 1992 | pmid = 1358792 | doi = 10.1016/S0888-7543(05)80298-8 }}</ref> which has an important role in cholesterol metabolism. It is a [[cytochrome P450]] enzyme, which belongs to the [[oxidoreductase]] class, and converts cholesterol to [[7-alpha-hydroxycholesterol]], the first and rate limiting step in [[bile acid]] synthesis. |
||
The inhibition of cholesterol 7-alpha-hydroxylase (CYP7A1) represses bile acid biosynthesis.<ref>{{cite thesis | type = Ph.D. | last = Miao |first= Ji |
The inhibition of cholesterol 7-alpha-hydroxylase (CYP7A1) represses bile acid biosynthesis.<ref>{{cite thesis | type = Ph.D. | last = Miao | first = Ji | date = 2008 | title = Regulation of Bile Acid Biosynthesis by Orphan Nuclear Receptor Small Heterodimer Partner | publisher = University of Illinois at Urbana-Champaign | url = https://books.google.com/books?id=PFj5fKLbwI0C&q=cholesterol+7+alpha+hydroxylase&pg=PA23 }}{{Dead link|date=March 2024 |bot=InternetArchiveBot |fix-attempted=yes }}</ref> |
||
== Evolution == |
== Evolution == |
||
Sequence comparisons indicated a huge similarity between cytochromes P450 identified in man and bacteria, and suggested that the superfamily |
Sequence comparisons indicated a huge similarity between cytochromes P450 identified in man and bacteria, and suggested that the superfamily cytochrome P450 first originated from a common ancestral gene some three billion years ago. |
||
The superfamily |
The superfamily cytochrome P450 was named in 1961, because of the 450-nm spectral peak pigment that cytochrome P450 has when reduced and bound to carbon monoxide. In the early 1960s, P450 was thought to be one enzyme, and by the mid 1960s it was associated with drug and steroid metabolism.<ref name="Nebert_2002">{{cite journal | vauthors = Nebert DW, Russell DW | s2cid = 13577054 | title = Clinical importance of the cytochromes P450 | journal = Lancet | volume = 360 | issue = 9340 | pages = 1155–62 | year = 2002 | pmid = 12387968 | doi = 10.1016/S0140-6736(02)11203-7 }}</ref> |
||
However, the membrane-associated and hydrophobic nature of the enzyme system impeded purification, and the number of proteins involved could not be accurately counted. Advances in mRNA purification in the early 1980s allowed to isolate the first cDNA encoding a complete cytochrome P450 (CYP) protein, and thereafter, results of many cloning studies have revealed a large number of different enzymes.<ref name="Nebert_2002" /> |
However, the membrane-associated and hydrophobic nature of the enzyme system impeded purification, and the number of proteins involved could not be accurately counted. Advances in mRNA purification in the early 1980s allowed to isolate the first cDNA encoding a complete cytochrome P450 (CYP) protein, and thereafter, results of many cloning studies have revealed a large number of different enzymes.<ref name="Nebert_2002" /> |
||
| Line 22: | Line 24: | ||
== Molecular structure == |
== Molecular structure == |
||
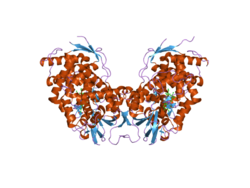
Cholesterol 7 alpha hydroxylase consists of 491 [[amino acids]], which on folding forms 23 [[alpha helices]] and 26 [[beta sheet]]s.<ref name="pmid19346330">{{cite journal |author = Chiang JY|title = Bile acids: regulation of synthesis|journal = J. Lipid Res.|volume = 50|issue = 10|pages = 1955–66|date = October 2009|pmid = 19346330|pmc = 2739756|doi = 10.1194/jlr.R900010-JLR200 |
Cholesterol 7 alpha hydroxylase consists of 491 [[amino acids]], which on folding forms 23 [[alpha helices]] and 26 [[beta sheet]]s.<ref name="pmid19346330">{{cite journal |author = Chiang JY|title = Bile acids: regulation of synthesis|journal = J. Lipid Res.|volume = 50|issue = 10|pages = 1955–66|date = October 2009|pmid = 19346330|pmc = 2739756|doi = 10.1194/jlr.R900010-JLR200 |doi-access=free }}</ref><ref>{{cite web|title = RCSB PDB|url = http://www.rcsb.org/pdb/results/results-sp.php|website = RCSB PDB|access-date = 2015-10-18}}{{Dead link|date=July 2019 |bot=InternetArchiveBot |fix-attempted=yes }}</ref> |
||
[[File:Cholesterol 7- |
[[File:Cholesterol 7-alpha Hydroxylase Rotation.gif|centre|thumb|178x178px|Cholesterol 7-alpha Hydroxylase Rotation]] |
||
== Function == |
== Function == |
||
Cholesterol 7 alpha-hydroxylase is a [[cytochrome P450]] [[heme]] [[enzyme]] that oxidizes cholesterol in the position 7 using [[molecular]] [[oxygen]]. It is an oxidoreductase. CYP7A1 is located in the [[endoplasmic reticulum]] (ER) and is important for the synthesis of bile acid and the regulation of cholesterol levels.<ref name="pmid19346330"/><ref>{{ |
Cholesterol 7 alpha-hydroxylase is a [[cytochrome P450]] [[heme]] [[enzyme]] that oxidizes cholesterol in the position 7 using [[molecular]] [[oxygen]]. It is an oxidoreductase. CYP7A1 is located in the [[endoplasmic reticulum]] (ER) and is important for the synthesis of bile acid and the regulation of cholesterol levels.<ref name="pmid19346330"/><ref>{{cite web|title = Síntesis de Ácido Biliar, el Metabolismo y las Funciones Biológicas|url = http://themedicalbiochemistrypage.org/es/bileacids-sp.php|access-date = 2015-10-15}}</ref>[[File:Hydrophobic structure of Cholesterol 7-alpha-hydroxylase.jpg|thumb|Hydrophobic image of cholesterol 7-alpha-hydroxylase|317x317px]] |
||
=== Synthesis of bile acid === |
=== Synthesis of bile acid === |
||
| Line 32: | Line 34: | ||
Cholesterol 7 alpha-hydroxylase is the [[rate-limiting enzyme]] in the synthesis of [[bile acid]] from [[cholesterol]] via the classic pathway, catalyzing the formation of [[7α-hydroxycholesterol]]. The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients.<ref name="pmid19346330" /> |
Cholesterol 7 alpha-hydroxylase is the [[rate-limiting enzyme]] in the synthesis of [[bile acid]] from [[cholesterol]] via the classic pathway, catalyzing the formation of [[7α-hydroxycholesterol]]. The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients.<ref name="pmid19346330" /> |
||
Bile acids have powerful toxic properties like membrane disruption and there are a wide range of mechanisms to restrict their accumulation in tissues and blood. The discovery of [[farnesoid X receptor]] (FXR) which is located in the liver, has opened new insights. Bile acid activation of FXR represses the expression of CYP7A1 via, raising the expression of |
Bile acids have powerful toxic properties like membrane disruption and there are a wide range of mechanisms to restrict their accumulation in tissues and blood. The discovery of [[farnesoid X receptor]] (FXR) which is located in the liver, has opened new insights. Bile acid activation of FXR represses the expression of CYP7A1 via, raising the expression of [[small heterodimer partner]] (SHP, NR0B2), a non-DNA binding protein.<ref name="pmid19346330" /> |
||
[[File:Atomic structure of cholesterol 7-alpha-hydroxylase.jpeg|thumb|Atomic structure of cholesterol 7-alpha-hydroxylase|317x317px]] |
|||
The increased abundance of SHP causes it to associate with liver receptor homolog (LRH)-1, an obligate factor required for the transcription of CYP7A1. Furthermore, there is an "FXR/SHP-independent" mechanism that also represses CYP7A1 expression. This "FXR/SHP-independent" pathway involves the interaction of bile acids with liver macrophages, which finally induces the expression and secretion of cytokines. These inflammatory cytokines, which include tumor necrosis factor alpha and interleukin-1beta, act upon the liver parenchymal cells causing a rapid repression of the CYP7A1 gene.<ref name="pmid19346330" /> |
|||
=== Regulation of activity === |
=== Regulation of activity === |
||
| Line 38: | Line 43: | ||
Regulation of CYP7A1 occurs at several levels including synthesis. Bile acids, steroid hormones, inflammatory cytokines, insulin, and growth factors inhibit CYP7A1 transcription through the 5′-upstream region of the promoter.<ref name="pmid19346330" /> The average life of this enzyme is between two and three hours. Activity can be regulated by phosphorylation-dephosphorylation. |
Regulation of CYP7A1 occurs at several levels including synthesis. Bile acids, steroid hormones, inflammatory cytokines, insulin, and growth factors inhibit CYP7A1 transcription through the 5′-upstream region of the promoter.<ref name="pmid19346330" /> The average life of this enzyme is between two and three hours. Activity can be regulated by phosphorylation-dephosphorylation. |
||
CYP7A1 is upregulated by the nuclear receptor [[Liver X receptor|LXR]] (liver X receptor) when cholesterol (to be specific, oxysterol) levels are high.<ref name="pmid11051540">{{cite journal | vauthors = Chawla A, Saez E, Evans RM | title = Don't know much bile-ology | journal = Cell | volume = 103 | issue = 1 | pages = 1–4 | date = Sep 2000 | pmid = 11051540 | doi = 10.1016/S0092-8674(00)00097-0 }}</ref> The effect of this upregulation is to increase the production of bile acids and reduce the level of cholesterol in hepatocytes. |
CYP7A1 is upregulated by the nuclear receptor [[Liver X receptor|LXR]] (liver X receptor) when cholesterol (to be specific, oxysterol) levels are high.<ref name="pmid11051540">{{cite journal | vauthors = Chawla A, Saez E, Evans RM | s2cid = 17408369 | title = Don't know much bile-ology | journal = Cell | volume = 103 | issue = 1 | pages = 1–4 | date = Sep 2000 | pmid = 11051540 | doi = 10.1016/S0092-8674(00)00097-0 | doi-access = free }}</ref> The effect of this upregulation is to increase the production of bile acids and reduce the level of cholesterol in hepatocytes. |
||
It is [[downregulated]] by [[ |
It is [[downregulated]] by [[sterol regulatory element-binding protein]]s (SREBP) when plasma cholesterol levels are low. |
||
Bile acids provide feedback inhibition of CYP7A1 by at least two different pathways, both involving the |
Bile acids provide feedback inhibition of CYP7A1 by at least two different pathways, both involving the farnesoid X receptor, FXR.<ref name="pmid19346330" /> In the liver, bile acids bound to FXR induce [[small heterodimer partner]], SHP which binds to LRH-1 and so inhibits the transcription of the enzyme. In the intestine, bile acids/FXR stimulate production of [[FGF15/19]] (depending on species), which then acts as a hormone in the liver via [[FGFR4]].<ref name="pmid19346330" /> |
||
== Enzymatic mechanism == |
== Enzymatic mechanism == |
||
| Line 58: | Line 63: | ||
== Clinical significance == |
== Clinical significance == |
||
Deficiency of this enzyme will increase the possibility of cholesterol gallstones.<ref name="pmid1682550">{{cite journal | vauthors = Paumgartner G, Sauerbruch T | title = Gallstones: pathogenesis | journal = Lancet | volume = 338 | issue = 8775 | pages = 1117–21 | date = Nov 1991 | pmid = 1682550 | doi = 10.1016/0140-6736(91)91972-W }}</ref> |
Deficiency of this enzyme will increase the possibility of cholesterol gallstones.<ref name="pmid1682550">{{cite journal | vauthors = Paumgartner G, Sauerbruch T | s2cid = 205037880 | title = Gallstones: pathogenesis | journal = Lancet | volume = 338 | issue = 8775 | pages = 1117–21 | date = Nov 1991 | pmid = 1682550 | doi = 10.1016/0140-6736(91)91972-W }}</ref> |
||
Disruption of CYP7A1 from classic bile acid synthesis in mice leads to either increased postnatal death or a milder phenotype with elevated serum cholesterol.<ref name="pmid11051540" /> The latter is similar to the case in humans, where CYP7A1 mutations associate with high plasma low-density lipoprotein and hepatic cholesterol content, as well as deficient bile acid excretion. There is also a synergy between plasma low-density lipoprotein cholesterol (LDL-C) and risks of [[coronary artery disease]] (CAD).<ref name="pmid11051540" /> Glucose signaling also induces CYP7A1 gene transcription by epigenetic regulation of the histone acetylation status. Glucose induction of bile acid synthesis have an important implication in metabolic control of glucose, lipid, and energy homeostasis under normal and diabetic conditions.<ref>{{cite journal | vauthors = Li T, Chanda D, Zhang Y, Choi HS, Chiang JY | title = Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes | journal = Journal of Lipid Research | volume = 51 | issue = 4 | pages = 832–42 | date = Apr 2010 | pmid = 19965590 | pmc = 2842145 | doi = 10.1194/jlr.M002782 }}</ref> CYP7A1-rs3808607 and APOE isoform are associated with the extent of reduction in circulating LDL cholesterol in response to |
Disruption of CYP7A1 from classic bile acid synthesis in mice leads to either increased postnatal death or a milder phenotype with elevated serum cholesterol.<ref name="pmid11051540" /> The latter is similar to the case in humans, where CYP7A1 mutations associate with high plasma low-density lipoprotein and hepatic cholesterol content, as well as deficient bile acid excretion. There is also a synergy between plasma [[low-density lipoprotein cholesterol]] (LDL-C) and risks of [[coronary artery disease]] (CAD).<ref name="pmid11051540" /> Glucose signaling also induces CYP7A1 gene transcription by [[epigenetic regulation]] of the [[histone acetylation]] status. Glucose induction of bile acid synthesis have an important implication in metabolic control of glucose, lipid, and energy homeostasis under normal and diabetic conditions.<ref>{{cite journal | vauthors = Li T, Chanda D, Zhang Y, Choi HS, Chiang JY | title = Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes | journal = Journal of Lipid Research | volume = 51 | issue = 4 | pages = 832–42 | date = Apr 2010 | pmid = 19965590 | pmc = 2842145 | doi = 10.1194/jlr.M002782 |doi-access=free }}</ref> CYP7A1-rs3808607 and [[apolipoprotein E]] (APOE) isoform are associated with the extent of reduction in circulating LDL cholesterol in response to [[phytosterol|plant sterol]] consumption and could serve as potential predictive genetic markers to identify individuals who would derive maximum LDL cholesterol lowering with plant sterol consumption.<ref name="pmid26333513">{{cite journal | vauthors = MacKay DS, Eck PK, Gebauer SK, Baer DJ, Jones PJ | title = CYP7A1-rs3808607 and APOE isoform associate with LDL cholesterol lowering after plant sterol consumption in a randomized clinical trial | journal = The American Journal of Clinical Nutrition | volume = 102 | issue = 4 | pages = 951–7 | date = Oct 2015 | pmid = 26333513 | doi = 10.3945/ajcn.115.109231 | doi-access = free }}</ref> Genetic variations in CYP7A1 influence its expression and thus may affect the risk of gallstone disease and gallbladder cancer.<ref name="pmid20005541">{{cite journal | vauthors = Srivastava A, Choudhuri G, Mittal B | title = CYP7A1 (-204 A>C; rs3808607 and -469 T>C; rs3824260) promoter polymorphisms and risk of gallbladder cancer in North Indian population | journal = Metab. Clin. Exp. | volume = 59 | issue = 6 | pages = 767–73 | year = 2010 | pmid = 20005541 | doi = 10.1016/j.metabol.2009.09.021 }}</ref> |
||
One of the many lipid lowering effects of the [[fibrate]] drug class is mediated through the inhibition of transcription of this enzyme.<ref>{{cite journal | vauthors = Gbaguidi GF, Agellon LB | title = The inhibition of the human cholesterol 7alpha-hydroxylase gene (CYP7A1) promoter by fibrates in cultured cells is mediated via the liver x receptor alpha and peroxisome proliferator-activated receptor alpha heterodimer | journal = Nucleic Acids Research | volume = 32 | issue = 3 | pages = 1113–21 | date = 2004-01-01 | pmid = 14960721 | pmc = 373396 | doi = 10.1093/nar/gkh260 }}</ref> This inhibition leads to more cholesterol in the bile, which is the body's only route of cholesterol excretion. This also increases the risk of [[Gallstone|cholesterol gallstone]] formation. |
One of the many lipid lowering effects of the [[fibrate]] drug class is mediated through the inhibition of transcription of this enzyme.<ref>{{cite journal | vauthors = Gbaguidi GF, Agellon LB | title = The inhibition of the human cholesterol 7alpha-hydroxylase gene (CYP7A1) promoter by fibrates in cultured cells is mediated via the liver x receptor alpha and peroxisome proliferator-activated receptor alpha heterodimer | journal = Nucleic Acids Research | volume = 32 | issue = 3 | pages = 1113–21 | date = 2004-01-01 | pmid = 14960721 | pmc = 373396 | doi = 10.1093/nar/gkh260 }}</ref> This inhibition leads to more cholesterol in the bile, which is the body's only route of cholesterol excretion. This also increases the risk of [[Gallstone|cholesterol gallstone]] formation. |
||
Inhibition of CYP7A1 is thought to be involved in or responsible for the [[hepatotoxicity]] associated with [[ketoconazole]].<ref name="Cuevas-RamosLim2016">{{cite journal|last1=Cuevas-Ramos|first1=Daniel|last2=Lim|first2=Dawn Shao Ting|last3=Fleseriu|first3=Maria|title=Update on medical treatment for Cushing's disease|journal=Clinical Diabetes and Endocrinology|volume=2|issue=1|pages=16|year=2016|issn=2055-8260|doi=10.1186/s40842-016-0033-9|pmid=28702250|pmc=5471955}}</ref> The [[levorotatory enantiomer]] of ketoconazole, [[levoketoconazole]], shows 12-fold reduced [[potency (pharmacology)|potency]] in inhibition of this enzyme, and is under development for certain indications (e.g., [[Cushing's syndrome]]) as a replacement for ketoconazole with reduced [[toxicity]] and improved [[tolerability]] and [[drug safety|safety]].<ref name="Cuevas-RamosLim2016" /> |
Inhibition of CYP7A1 is thought to be involved in or responsible for the [[hepatotoxicity]] associated with [[ketoconazole]].<ref name="Cuevas-RamosLim2016">{{cite journal|last1=Cuevas-Ramos|first1=Daniel|last2=Lim|first2=Dawn Shao Ting|last3=Fleseriu|first3=Maria|title=Update on medical treatment for Cushing's disease|journal=Clinical Diabetes and Endocrinology|volume=2|issue=1|pages=16|year=2016|issn=2055-8260|doi=10.1186/s40842-016-0033-9|pmid=28702250|pmc=5471955 |doi-access=free }}</ref> The [[levorotatory enantiomer]] of ketoconazole, [[levoketoconazole]], shows 12-fold reduced [[potency (pharmacology)|potency]] in inhibition of this enzyme, and is under development for certain indications (e.g., [[Cushing's syndrome]]) as a replacement for ketoconazole with reduced [[toxicity]] and improved [[tolerability]] and [[drug safety|safety]].<ref name="Cuevas-RamosLim2016" /> |
||
==See also== |
==See also== |
||
| ⚫ | |||
* [[Steroidogenic enzyme]] |
* [[Steroidogenic enzyme]] |
||
== References == |
== References == |
||
{{ |
{{reflist}} |
||
== Further reading == |
== Further reading == |
||
{{refbegin|33em}} |
{{refbegin|33em}} |
||
* {{cite journal | vauthors = Davis RA, Miyake JH, Hui TY, Spann NJ | title = Regulation of cholesterol-7alpha-hydroxylase: BAREly missing a SHP | journal = Journal of Lipid Research | volume = 43 | issue = 4 | pages = 533–43 | date = Apr 2002 | pmid = 11907135 | doi = }} |
* {{cite journal | vauthors = Davis RA, Miyake JH, Hui TY, Spann NJ | title = Regulation of cholesterol-7alpha-hydroxylase: BAREly missing a SHP | journal = Journal of Lipid Research | volume = 43 | issue = 4 | pages = 533–43 | date = Apr 2002 | doi = 10.1016/S0022-2275(20)31482-6 | pmid = 11907135 | doi-access = free }} |
||
* {{cite journal | vauthors = Kim HJ, Park HY, Kim E, Lee KS, Kim KK, Choi BO, Kim SM, Bae JS, Lee SO, Chun JY, Park TJ, Cheong HS, Jo I, Shin HD | title = Common CYP7A1 promoter polymorphism associated with risk of neuromyelitis optica | journal = Neurobiology of Disease | volume = 37 | issue = 2 | pages = 349–55 | date = Feb 2010 | pmid = 19850125 | doi = 10.1016/j.nbd.2009.10.013 }} |
* {{cite journal | vauthors = Kim HJ, Park HY, Kim E, Lee KS, Kim KK, Choi BO, Kim SM, Bae JS, Lee SO, Chun JY, Park TJ, Cheong HS, Jo I, Shin HD | s2cid = 40067459 | title = Common CYP7A1 promoter polymorphism associated with risk of neuromyelitis optica | journal = Neurobiology of Disease | volume = 37 | issue = 2 | pages = 349–55 | date = Feb 2010 | pmid = 19850125 | doi = 10.1016/j.nbd.2009.10.013 }} |
||
* {{cite journal | vauthors = Holmes MV, Shah T, Vickery C, Smeeth L, Hingorani AD, Casas JP | title = Fulfilling the promise of personalized medicine? Systematic review and field synopsis of pharmacogenetic studies | journal = PLOS ONE | volume = 4 | issue = 12 | pages = e7960 | year = 2009 | pmid = 19956635 | pmc = 2778625 | doi = 10.1371/journal.pone.0007960 | editor1-last = Luo | editor1-first = Yuan | |
* {{cite journal | vauthors = Holmes MV, Shah T, Vickery C, Smeeth L, Hingorani AD, Casas JP | title = Fulfilling the promise of personalized medicine? Systematic review and field synopsis of pharmacogenetic studies | journal = PLOS ONE | volume = 4 | issue = 12 | pages = e7960 | year = 2009 | pmid = 19956635 | pmc = 2778625 | doi = 10.1371/journal.pone.0007960 | bibcode = 2009PLoSO...4.7960H | editor1-last = Luo | editor1-first = Yuan | doi-access = free }} |
||
* {{cite journal | vauthors = Lipkin SM, Chao EC, Moreno V, Rozek LS, Rennert H, Pinchev M, Dizon D, Rennert G, Kopelovich L, Gruber SB | title = Genetic variation in 3-hydroxy-3-methylglutaryl CoA reductase modifies the chemopreventive activity of statins for colorectal cancer | journal = Cancer Prevention Research | volume = 3 | issue = 5 | pages = 597–603 | date = May 2010 | pmid = 20403997 | doi = 10.1158/1940-6207.CAPR-10-0007 }} |
* {{cite journal | vauthors = Lipkin SM, Chao EC, Moreno V, Rozek LS, Rennert H, Pinchev M, Dizon D, Rennert G, Kopelovich L, Gruber SB | title = Genetic variation in 3-hydroxy-3-methylglutaryl CoA reductase modifies the chemopreventive activity of statins for colorectal cancer | journal = Cancer Prevention Research | volume = 3 | issue = 5 | pages = 597–603 | date = May 2010 | pmid = 20403997 | doi = 10.1158/1940-6207.CAPR-10-0007 | doi-access = free | url = https://aacr.figshare.com/articles/journal_contribution/Supplementary_Tables_1-2_from_Genetic_Variation_in_3-Hydroxy-3-Methylglutaryl_CoA_Reductase_Modifies_the_Chemopreventive_Activity_of_Statins_for_Colorectal_Cancer/22524390/1/files/39987216.pdf }} |
||
* {{cite journal | vauthors = Lambrinoudaki I, Kaparos G, Rizos D, Galapi F, Alexandrou A, Sergentanis TN, Creatsa M, Christodoulakos G, Kouskouni E, Botsis D | title = Apolipoprotein E and paraoxonase 1 polymorphisms are associated with lower serum thyroid hormones in postmenopausal women | journal = Clinical Endocrinology | volume = 71 | issue = 2 | pages = 284–90 | date = Aug 2009 | pmid = 19018779 | doi = 10.1111/j.1365-2265.2008.03476.x }} |
* {{cite journal | vauthors = Lambrinoudaki I, Kaparos G, Rizos D, Galapi F, Alexandrou A, Sergentanis TN, Creatsa M, Christodoulakos G, Kouskouni E, Botsis D | title = Apolipoprotein E and paraoxonase 1 polymorphisms are associated with lower serum thyroid hormones in postmenopausal women | journal = Clinical Endocrinology | volume = 71 | issue = 2 | pages = 284–90 | date = Aug 2009 | pmid = 19018779 | doi = 10.1111/j.1365-2265.2008.03476.x | s2cid = 205284974 }} |
||
* {{cite journal | vauthors = Poduri A, Khullar M, Bahl A, Sharma YP, Talwar KK | title = A combination of proatherogenic single-nucleotide polymorphisms is associated with increased risk of coronary artery disease and myocardial infarction in Asian Indians | journal = DNA and Cell Biology | volume = 28 | issue = 9 | pages = 451–60 | date = Sep 2009 | pmid = 19558216 | doi = 10.1089/dna.2009.0887 }} |
* {{cite journal | vauthors = Poduri A, Khullar M, Bahl A, Sharma YP, Talwar KK | title = A combination of proatherogenic single-nucleotide polymorphisms is associated with increased risk of coronary artery disease and myocardial infarction in Asian Indians | journal = DNA and Cell Biology | volume = 28 | issue = 9 | pages = 451–60 | date = Sep 2009 | pmid = 19558216 | doi = 10.1089/dna.2009.0887 }} |
||
* {{cite journal | vauthors = Li T, Chanda D, Zhang Y, Choi HS, Chiang JY | title = Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes | journal = Journal of Lipid Research | volume = 51 | issue = 4 | pages = 832–42 | date = Apr 2010 | pmid = 19965590 | pmc = 2842145 | doi = 10.1194/jlr.M002782 }} |
* {{cite journal | vauthors = Li T, Chanda D, Zhang Y, Choi HS, Chiang JY | title = Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes | journal = Journal of Lipid Research | volume = 51 | issue = 4 | pages = 832–42 | date = Apr 2010 | pmid = 19965590 | pmc = 2842145 | doi = 10.1194/jlr.M002782 |doi-access=free }} |
||
* {{cite journal | vauthors = Kovár J, Lenícek M, Zimolová M, Vítek L, Jirsa M, Pitha J | title = Regulation of diurnal variation of cholesterol 7alpha-hydroxylase (CYP7A1) activity in healthy subjects | journal = Physiological Research | volume = 59 | issue = 2 | pages = 233–8 | year = 2010 | pmid = 19537927 | doi = }} |
* {{cite journal | vauthors = Kovár J, Lenícek M, Zimolová M, Vítek L, Jirsa M, Pitha J | title = Regulation of diurnal variation of cholesterol 7alpha-hydroxylase (CYP7A1) activity in healthy subjects | journal = Physiological Research | volume = 59 | issue = 2 | pages = 233–8 | year = 2010 | doi = 10.33549/physiolres.931753 | pmid = 19537927 | doi-access = free }} |
||
* {{cite journal | vauthors = Chien KL, Wang KC, Chen YC, Chao CL, Hsu HC, Chen MF, Chen WJ | title = Common sequence variants in pharmacodynamic and pharmacokinetic pathway-related genes conferring LDL cholesterol response to statins | journal = Pharmacogenomics | volume = 11 | issue = 3 | pages = 309–17 | date = Mar 2010 | pmid = 20235787 | doi = 10.2217/pgs.09.160 }} |
* {{cite journal | vauthors = Chien KL, Wang KC, Chen YC, Chao CL, Hsu HC, Chen MF, Chen WJ | title = Common sequence variants in pharmacodynamic and pharmacokinetic pathway-related genes conferring LDL cholesterol response to statins | journal = Pharmacogenomics | volume = 11 | issue = 3 | pages = 309–17 | date = Mar 2010 | pmid = 20235787 | doi = 10.2217/pgs.09.160 }} |
||
* {{cite journal | vauthors = Saito A, Kawamoto M, Kamatani N | title = Association study between single-nucleotide polymorphisms in 199 drug-related genes and commonly measured quantitative traits of 752 healthy Japanese subjects | journal = Journal of Human Genetics | volume = 54 | issue = 6 | pages = 317–23 | date = Jun 2009 | pmid = 19343046 | doi = 10.1038/jhg.2009.31 }} |
* {{cite journal | vauthors = Saito A, Kawamoto M, Kamatani N | title = Association study between single-nucleotide polymorphisms in 199 drug-related genes and commonly measured quantitative traits of 752 healthy Japanese subjects | journal = Journal of Human Genetics | volume = 54 | issue = 6 | pages = 317–23 | date = Jun 2009 | pmid = 19343046 | doi = 10.1038/jhg.2009.31 | doi-access = free }} |
||
* {{cite journal | vauthors = Nelson DR, Zeldin DC, Hoffman SM, Maltais LJ, Wain HM, Nebert DW | title = Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants | journal = Pharmacogenetics | volume = 14 | issue = 1 | pages = 1–18 | date = Jan 2004 | pmid = 15128046 | doi = 10.1097/00008571-200401000-00001 |
* {{cite journal | vauthors = Nelson DR, Zeldin DC, Hoffman SM, Maltais LJ, Wain HM, Nebert DW | s2cid = 18448751 | title = Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants | journal = Pharmacogenetics | volume = 14 | issue = 1 | pages = 1–18 | date = Jan 2004 | pmid = 15128046 | doi = 10.1097/00008571-200401000-00001 }} |
||
* {{cite journal | vauthors = Li T, Ma H, Park YJ, Lee YK, Strom S, Moore DD, Chiang JY | title = Forkhead box transcription factor O1 inhibits cholesterol 7alpha-hydroxylase in human hepatocytes and in high fat diet-fed mice | journal = Biochimica et Biophysica Acta | volume = 1791 | issue = 10 | pages = 991–6 | date = Oct 2009 | pmid = 19463968 | pmc = 2743791 | doi = 10.1016/j.bbalip.2009.05.004 }} |
* {{cite journal | vauthors = Li T, Ma H, Park YJ, Lee YK, Strom S, Moore DD, Chiang JY | title = Forkhead box transcription factor O1 inhibits cholesterol 7alpha-hydroxylase in human hepatocytes and in high fat diet-fed mice | journal = Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids | volume = 1791 | issue = 10 | pages = 991–6 | date = Oct 2009 | pmid = 19463968 | pmc = 2743791 | doi = 10.1016/j.bbalip.2009.05.004 }} |
||
* {{cite journal | vauthors = Chakrabarti B, Dudbridge F, Kent L, Wheelwright S, Hill-Cawthorne G, Allison C, Banerjee-Basu S, Baron-Cohen S | title = Genes related to sex steroids, neural growth, and social-emotional behavior are associated with autistic traits, empathy, and Asperger syndrome | journal = Autism Research | volume = 2 | issue = 3 | pages = 157–77 | date = Jun 2009 | pmid = 19598235 | doi = 10.1002/aur.80 }} |
* {{cite journal | vauthors = Chakrabarti B, Dudbridge F, Kent L, Wheelwright S, Hill-Cawthorne G, Allison C, Banerjee-Basu S, Baron-Cohen S | title = Genes related to sex steroids, neural growth, and social-emotional behavior are associated with autistic traits, empathy, and Asperger syndrome | journal = Autism Research | volume = 2 | issue = 3 | pages = 157–77 | date = Jun 2009 | pmid = 19598235 | doi = 10.1002/aur.80 | s2cid = 18239947 }} |
||
* {{cite journal | vauthors = Barcelos AL, Chies R, Almeida SE, Fiegenbaum M, Schweigert ID, Chula FG, Rossetti ML, Silva CM | title = Association of CYP7A1 -278A>C polymorphism and the response of plasma triglyceride after dietary intervention in dyslipidemic patients | journal = Brazilian Journal of Medical and Biological Research | volume = 42 | issue = 6 | pages = 487–93 | date = Jun 2009 | pmid = 19448895 | doi = 10.1590/s0100-879x2009000600003 }} |
* {{cite journal | vauthors = Barcelos AL, Chies R, Almeida SE, Fiegenbaum M, Schweigert ID, Chula FG, Rossetti ML, Silva CM | title = Association of CYP7A1 -278A>C polymorphism and the response of plasma triglyceride after dietary intervention in dyslipidemic patients | journal = Brazilian Journal of Medical and Biological Research | volume = 42 | issue = 6 | pages = 487–93 | date = Jun 2009 | pmid = 19448895 | doi = 10.1590/s0100-879x2009000600003 | doi-access = free }} |
||
* {{cite journal | vauthors = Voora D, Shah SH, Reed CR, Zhai J, Crosslin DR, Messer C, Salisbury BA, Ginsburg GS | title = Pharmacogenetic predictors of statin-mediated low-density lipoprotein cholesterol reduction and dose response | journal = Circulation: Cardiovascular Genetics | volume = 1 | issue = 2 | pages = 100–6 | date = Dec 2008 | pmid = 20031551 | pmc = 2995295 | doi = 10.1161/CIRCGENETICS.108.795013 }} |
* {{cite journal | vauthors = Voora D, Shah SH, Reed CR, Zhai J, Crosslin DR, Messer C, Salisbury BA, Ginsburg GS | title = Pharmacogenetic predictors of statin-mediated low-density lipoprotein cholesterol reduction and dose response | journal = Circulation: Cardiovascular Genetics | volume = 1 | issue = 2 | pages = 100–6 | date = Dec 2008 | pmid = 20031551 | pmc = 2995295 | doi = 10.1161/CIRCGENETICS.108.795013 }} |
||
* {{cite journal | vauthors = Schwarz M, Lund EG, Russell DW | title = Two 7 alpha-hydroxylase enzymes in bile acid biosynthesis | journal = Current Opinion in Lipidology | volume = 9 | issue = 2 | pages = 113–8 | date = Apr 1998 | pmid = 9559267 | doi = 10.1097/00041433-199804000-00006 }} |
* {{cite journal | vauthors = Schwarz M, Lund EG, Russell DW | title = Two 7 alpha-hydroxylase enzymes in bile acid biosynthesis | journal = Current Opinion in Lipidology | volume = 9 | issue = 2 | pages = 113–8 | date = Apr 1998 | pmid = 9559267 | doi = 10.1097/00041433-199804000-00006 }} |
||
* {{cite journal | vauthors = Beigneux A, Hofmann AF, Young SG | title = Human CYP7A1 deficiency: progress and enigmas | journal = The Journal of Clinical Investigation | volume = 110 | issue = 1 | pages = 29–31 | date = Jul 2002 | pmid = 12093884 | pmc = 151039 | doi = 10.1172/JCI16076 }} |
* {{cite journal | vauthors = Beigneux A, Hofmann AF, Young SG | title = Human CYP7A1 deficiency: progress and enigmas | journal = The Journal of Clinical Investigation | volume = 110 | issue = 1 | pages = 29–31 | date = Jul 2002 | pmid = 12093884 | pmc = 151039 | doi = 10.1172/JCI16076 }} |
||
* {{cite journal | vauthors = Ruaño G, Bernene J, Windemuth A, Bower B, Wencker D, Seip RL, Kocherla M, Holford TR, Petit WA, Hanks S | title = Physiogenomic comparison of edema and BMI in patients receiving rosiglitazone or pioglitazone | journal = Clinica Chimica Acta; International Journal of Clinical Chemistry | volume = 400 | issue = 1–2 | pages = 48–55 | date = Feb 2009 | pmid = 18996102 | doi = 10.1016/j.cca.2008.10.009 }} |
* {{cite journal | vauthors = Ruaño G, Bernene J, Windemuth A, Bower B, Wencker D, Seip RL, Kocherla M, Holford TR, Petit WA, Hanks S | title = Physiogenomic comparison of edema and BMI in patients receiving rosiglitazone or pioglitazone | journal = Clinica Chimica Acta; International Journal of Clinical Chemistry | volume = 400 | issue = 1–2 | pages = 48–55 | date = Feb 2009 | pmid = 18996102 | doi = 10.1016/j.cca.2008.10.009 }} |
||
* {{cite journal | vauthors = Schaap FG, van der Gaag NA, Gouma DJ, Jansen PL | title = High expression of the bile salt-homeostatic hormone fibroblast growth factor 19 in the liver of patients with extrahepatic cholestasis | journal = Hepatology | volume = 49 | issue = 4 | pages = 1228–35 | date = Apr 2009 | pmid = 19185005 | doi = 10.1002/hep.22771 }} |
* {{cite journal | vauthors = Schaap FG, van der Gaag NA, Gouma DJ, Jansen PL | title = High expression of the bile salt-homeostatic hormone fibroblast growth factor 19 in the liver of patients with extrahepatic cholestasis | journal = Hepatology | volume = 49 | issue = 4 | pages = 1228–35 | date = Apr 2009 | pmid = 19185005 | doi = 10.1002/hep.22771 | s2cid = 5306097 | doi-access = free }} |
||
* {{cite journal | vauthors = Srivastava A, Choudhuri G, Mittal B | title = CYP7A1 (-204 A>C; rs3808607 and -469 T>C; rs3824260) promoter polymorphisms and risk of gallbladder cancer in North Indian population | journal = Metabolism | volume = 59 | issue = 6 | pages = 767–73 | date = Jun 2010 | pmid = 20005541 | doi = 10.1016/j.metabol.2009.09.021 }} |
* {{cite journal | vauthors = Srivastava A, Choudhuri G, Mittal B | title = CYP7A1 (-204 A>C; rs3808607 and -469 T>C; rs3824260) promoter polymorphisms and risk of gallbladder cancer in North Indian population | journal = Metabolism | volume = 59 | issue = 6 | pages = 767–73 | date = Jun 2010 | pmid = 20005541 | doi = 10.1016/j.metabol.2009.09.021 }} |
||
{{refend}} |
{{refend}} |
||
| Line 104: | Line 110: | ||
{{Oxygenases}} |
{{Oxygenases}} |
||
{{Enzymes}} |
{{Enzymes}} |
||
| ⚫ | |||
[[Category:EC 1.14.13]] |
[[Category:EC 1.14.13]] |
||
Latest revision as of 03:33, 10 March 2024
Cholesterol 7 alpha-hydroxylase also known as cholesterol 7-alpha-monooxygenase or cytochrome P450 7A1 (CYP7A1) is an enzyme that in humans is encoded by the CYP7A1 gene[5] which has an important role in cholesterol metabolism. It is a cytochrome P450 enzyme, which belongs to the oxidoreductase class, and converts cholesterol to 7-alpha-hydroxycholesterol, the first and rate limiting step in bile acid synthesis.
The inhibition of cholesterol 7-alpha-hydroxylase (CYP7A1) represses bile acid biosynthesis.[6]
Evolution[edit]
Sequence comparisons indicated a huge similarity between cytochromes P450 identified in man and bacteria, and suggested that the superfamily cytochrome P450 first originated from a common ancestral gene some three billion years ago.
The superfamily cytochrome P450 was named in 1961, because of the 450-nm spectral peak pigment that cytochrome P450 has when reduced and bound to carbon monoxide. In the early 1960s, P450 was thought to be one enzyme, and by the mid 1960s it was associated with drug and steroid metabolism.[7]
However, the membrane-associated and hydrophobic nature of the enzyme system impeded purification, and the number of proteins involved could not be accurately counted. Advances in mRNA purification in the early 1980s allowed to isolate the first cDNA encoding a complete cytochrome P450 (CYP) protein, and thereafter, results of many cloning studies have revealed a large number of different enzymes.[7]
Advances in molecular biology and genomics facilitated the biochemical characterisation of individual P450 enzymes:
- The cytochromes P450 act on many endogenous substrates, introducing oxidative, peroxidative, and reductive changes into small molecules of widely different chemical structures. Substrates identified to date include saturated and unsaturated fatty acids, eicosanoids, sterols and steroids, bile acids, vitamin D3 derivatives, retinoids, and uroporphyrinogens.[7]
- Many cytochrome P450 enzymes can metabolise various exogenous compounds including drugs, environmental chemicals and pollutants, and natural plant products.[7]
- Metabolism of foreign chemicals frequently results in successful detoxication of the irritant; However, the actions of P450 enzymes can also generate toxic metabolites that contribute to increased risks of cancer, birth defects, and other toxic effects.
- The expression of many P450 enzymes is often induced by accumulation of a substrate.
- The ability of one P450 substrate to affect the concentrations of another in this manner is the basis for so-called drug-drug interactions, which complicate treatment.[7]
Molecular structure[edit]
Cholesterol 7 alpha hydroxylase consists of 491 amino acids, which on folding forms 23 alpha helices and 26 beta sheets.[8][9]

Function[edit]
Cholesterol 7 alpha-hydroxylase is a cytochrome P450 heme enzyme that oxidizes cholesterol in the position 7 using molecular oxygen. It is an oxidoreductase. CYP7A1 is located in the endoplasmic reticulum (ER) and is important for the synthesis of bile acid and the regulation of cholesterol levels.[8][10]

Synthesis of bile acid[edit]
Cholesterol 7 alpha-hydroxylase is the rate-limiting enzyme in the synthesis of bile acid from cholesterol via the classic pathway, catalyzing the formation of 7α-hydroxycholesterol. The unique detergent properties of bile acids are essential for the digestion and intestinal absorption of hydrophobic nutrients.[8]
Bile acids have powerful toxic properties like membrane disruption and there are a wide range of mechanisms to restrict their accumulation in tissues and blood. The discovery of farnesoid X receptor (FXR) which is located in the liver, has opened new insights. Bile acid activation of FXR represses the expression of CYP7A1 via, raising the expression of small heterodimer partner (SHP, NR0B2), a non-DNA binding protein.[8]

The increased abundance of SHP causes it to associate with liver receptor homolog (LRH)-1, an obligate factor required for the transcription of CYP7A1. Furthermore, there is an "FXR/SHP-independent" mechanism that also represses CYP7A1 expression. This "FXR/SHP-independent" pathway involves the interaction of bile acids with liver macrophages, which finally induces the expression and secretion of cytokines. These inflammatory cytokines, which include tumor necrosis factor alpha and interleukin-1beta, act upon the liver parenchymal cells causing a rapid repression of the CYP7A1 gene.[8]
Regulation of activity[edit]
Regulation of CYP7A1 occurs at several levels including synthesis. Bile acids, steroid hormones, inflammatory cytokines, insulin, and growth factors inhibit CYP7A1 transcription through the 5′-upstream region of the promoter.[8] The average life of this enzyme is between two and three hours. Activity can be regulated by phosphorylation-dephosphorylation.
CYP7A1 is upregulated by the nuclear receptor LXR (liver X receptor) when cholesterol (to be specific, oxysterol) levels are high.[11] The effect of this upregulation is to increase the production of bile acids and reduce the level of cholesterol in hepatocytes.
It is downregulated by sterol regulatory element-binding proteins (SREBP) when plasma cholesterol levels are low.
Bile acids provide feedback inhibition of CYP7A1 by at least two different pathways, both involving the farnesoid X receptor, FXR.[8] In the liver, bile acids bound to FXR induce small heterodimer partner, SHP which binds to LRH-1 and so inhibits the transcription of the enzyme. In the intestine, bile acids/FXR stimulate production of FGF15/19 (depending on species), which then acts as a hormone in the liver via FGFR4.[8]
Enzymatic mechanism[edit]
Specificity[edit]
One feature of enzymes is their high specificity. They are specific on a singular substrate, reaction or both together, that means, that the enzymes can catalyze all reactions wherein the substrate can experience.
The enzyme cholesterol 7 alpha hydroxylase catalyzes the reaction that converts cholesterol into cholesterol 7 alpha hydroxylase reducing and oxidizing that molecule.[8][12]
Interactive pathway map[edit]
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
Clinical significance[edit]
Deficiency of this enzyme will increase the possibility of cholesterol gallstones.[13]
Disruption of CYP7A1 from classic bile acid synthesis in mice leads to either increased postnatal death or a milder phenotype with elevated serum cholesterol.[11] The latter is similar to the case in humans, where CYP7A1 mutations associate with high plasma low-density lipoprotein and hepatic cholesterol content, as well as deficient bile acid excretion. There is also a synergy between plasma low-density lipoprotein cholesterol (LDL-C) and risks of coronary artery disease (CAD).[11] Glucose signaling also induces CYP7A1 gene transcription by epigenetic regulation of the histone acetylation status. Glucose induction of bile acid synthesis have an important implication in metabolic control of glucose, lipid, and energy homeostasis under normal and diabetic conditions.[14] CYP7A1-rs3808607 and apolipoprotein E (APOE) isoform are associated with the extent of reduction in circulating LDL cholesterol in response to plant sterol consumption and could serve as potential predictive genetic markers to identify individuals who would derive maximum LDL cholesterol lowering with plant sterol consumption.[15] Genetic variations in CYP7A1 influence its expression and thus may affect the risk of gallstone disease and gallbladder cancer.[16]
One of the many lipid lowering effects of the fibrate drug class is mediated through the inhibition of transcription of this enzyme.[17] This inhibition leads to more cholesterol in the bile, which is the body's only route of cholesterol excretion. This also increases the risk of cholesterol gallstone formation.
Inhibition of CYP7A1 is thought to be involved in or responsible for the hepatotoxicity associated with ketoconazole.[18] The levorotatory enantiomer of ketoconazole, levoketoconazole, shows 12-fold reduced potency in inhibition of this enzyme, and is under development for certain indications (e.g., Cushing's syndrome) as a replacement for ketoconazole with reduced toxicity and improved tolerability and safety.[18]
See also[edit]
References[edit]
- ^ a b c GRCh38: Ensembl release 89: ENSG00000167910 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000028240 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Cohen JC, Cali JJ, Jelinek DF, Mehrabian M, Sparkes RS, Lusis AJ, Russell DW, Hobbs HH (Sep 1992). "Cloning of the human cholesterol 7 alpha-hydroxylase gene (CYP7) and localization to chromosome 8q11-q12". Genomics. 14 (1): 153–61. doi:10.1016/S0888-7543(05)80298-8. PMID 1358792.
- ^ Miao J (2008). Regulation of Bile Acid Biosynthesis by Orphan Nuclear Receptor Small Heterodimer Partner (Ph.D.). University of Illinois at Urbana-Champaign.[permanent dead link]
- ^ a b c d e Nebert DW, Russell DW (2002). "Clinical importance of the cytochromes P450". Lancet. 360 (9340): 1155–62. doi:10.1016/S0140-6736(02)11203-7. PMID 12387968. S2CID 13577054.
- ^ a b c d e f g h i Chiang JY (October 2009). "Bile acids: regulation of synthesis". J. Lipid Res. 50 (10): 1955–66. doi:10.1194/jlr.R900010-JLR200. PMC 2739756. PMID 19346330.
- ^ "RCSB PDB". RCSB PDB. Retrieved 2015-10-18.[permanent dead link]
- ^ "Síntesis de Ácido Biliar, el Metabolismo y las Funciones Biológicas". Retrieved 2015-10-15.
- ^ a b c Chawla A, Saez E, Evans RM (Sep 2000). "Don't know much bile-ology". Cell. 103 (1): 1–4. doi:10.1016/S0092-8674(00)00097-0. PMID 11051540. S2CID 17408369.
- ^ Hedstrom L (2010). "Enzyme Specificity and Selectivity". eLS Citable Reviews in the Life Sciences. doi:10.1002/9780470015902.a0000716.pub2. ISBN 978-0470016176.
- ^ Paumgartner G, Sauerbruch T (Nov 1991). "Gallstones: pathogenesis". Lancet. 338 (8775): 1117–21. doi:10.1016/0140-6736(91)91972-W. PMID 1682550. S2CID 205037880.
- ^ Li T, Chanda D, Zhang Y, Choi HS, Chiang JY (Apr 2010). "Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes". Journal of Lipid Research. 51 (4): 832–42. doi:10.1194/jlr.M002782. PMC 2842145. PMID 19965590.
- ^ MacKay DS, Eck PK, Gebauer SK, Baer DJ, Jones PJ (Oct 2015). "CYP7A1-rs3808607 and APOE isoform associate with LDL cholesterol lowering after plant sterol consumption in a randomized clinical trial". The American Journal of Clinical Nutrition. 102 (4): 951–7. doi:10.3945/ajcn.115.109231. PMID 26333513.
- ^ Srivastava A, Choudhuri G, Mittal B (2010). "CYP7A1 (-204 A>C; rs3808607 and -469 T>C; rs3824260) promoter polymorphisms and risk of gallbladder cancer in North Indian population". Metab. Clin. Exp. 59 (6): 767–73. doi:10.1016/j.metabol.2009.09.021. PMID 20005541.
- ^ Gbaguidi GF, Agellon LB (2004-01-01). "The inhibition of the human cholesterol 7alpha-hydroxylase gene (CYP7A1) promoter by fibrates in cultured cells is mediated via the liver x receptor alpha and peroxisome proliferator-activated receptor alpha heterodimer". Nucleic Acids Research. 32 (3): 1113–21. doi:10.1093/nar/gkh260. PMC 373396. PMID 14960721.
- ^ a b Cuevas-Ramos D, Lim DS, Fleseriu M (2016). "Update on medical treatment for Cushing's disease". Clinical Diabetes and Endocrinology. 2 (1): 16. doi:10.1186/s40842-016-0033-9. ISSN 2055-8260. PMC 5471955. PMID 28702250.
Further reading[edit]
- Davis RA, Miyake JH, Hui TY, Spann NJ (Apr 2002). "Regulation of cholesterol-7alpha-hydroxylase: BAREly missing a SHP". Journal of Lipid Research. 43 (4): 533–43. doi:10.1016/S0022-2275(20)31482-6. PMID 11907135.
- Kim HJ, Park HY, Kim E, Lee KS, Kim KK, Choi BO, Kim SM, Bae JS, Lee SO, Chun JY, Park TJ, Cheong HS, Jo I, Shin HD (Feb 2010). "Common CYP7A1 promoter polymorphism associated with risk of neuromyelitis optica". Neurobiology of Disease. 37 (2): 349–55. doi:10.1016/j.nbd.2009.10.013. PMID 19850125. S2CID 40067459.
- Holmes MV, Shah T, Vickery C, Smeeth L, Hingorani AD, Casas JP (2009). Luo Y (ed.). "Fulfilling the promise of personalized medicine? Systematic review and field synopsis of pharmacogenetic studies". PLOS ONE. 4 (12): e7960. Bibcode:2009PLoSO...4.7960H. doi:10.1371/journal.pone.0007960. PMC 2778625. PMID 19956635.
- Lipkin SM, Chao EC, Moreno V, Rozek LS, Rennert H, Pinchev M, Dizon D, Rennert G, Kopelovich L, Gruber SB (May 2010). "Genetic variation in 3-hydroxy-3-methylglutaryl CoA reductase modifies the chemopreventive activity of statins for colorectal cancer" (PDF). Cancer Prevention Research. 3 (5): 597–603. doi:10.1158/1940-6207.CAPR-10-0007. PMID 20403997.
- Lambrinoudaki I, Kaparos G, Rizos D, Galapi F, Alexandrou A, Sergentanis TN, Creatsa M, Christodoulakos G, Kouskouni E, Botsis D (Aug 2009). "Apolipoprotein E and paraoxonase 1 polymorphisms are associated with lower serum thyroid hormones in postmenopausal women". Clinical Endocrinology. 71 (2): 284–90. doi:10.1111/j.1365-2265.2008.03476.x. PMID 19018779. S2CID 205284974.
- Poduri A, Khullar M, Bahl A, Sharma YP, Talwar KK (Sep 2009). "A combination of proatherogenic single-nucleotide polymorphisms is associated with increased risk of coronary artery disease and myocardial infarction in Asian Indians". DNA and Cell Biology. 28 (9): 451–60. doi:10.1089/dna.2009.0887. PMID 19558216.
- Li T, Chanda D, Zhang Y, Choi HS, Chiang JY (Apr 2010). "Glucose stimulates cholesterol 7alpha-hydroxylase gene transcription in human hepatocytes". Journal of Lipid Research. 51 (4): 832–42. doi:10.1194/jlr.M002782. PMC 2842145. PMID 19965590.
- Kovár J, Lenícek M, Zimolová M, Vítek L, Jirsa M, Pitha J (2010). "Regulation of diurnal variation of cholesterol 7alpha-hydroxylase (CYP7A1) activity in healthy subjects". Physiological Research. 59 (2): 233–8. doi:10.33549/physiolres.931753. PMID 19537927.
- Chien KL, Wang KC, Chen YC, Chao CL, Hsu HC, Chen MF, Chen WJ (Mar 2010). "Common sequence variants in pharmacodynamic and pharmacokinetic pathway-related genes conferring LDL cholesterol response to statins". Pharmacogenomics. 11 (3): 309–17. doi:10.2217/pgs.09.160. PMID 20235787.
- Saito A, Kawamoto M, Kamatani N (Jun 2009). "Association study between single-nucleotide polymorphisms in 199 drug-related genes and commonly measured quantitative traits of 752 healthy Japanese subjects". Journal of Human Genetics. 54 (6): 317–23. doi:10.1038/jhg.2009.31. PMID 19343046.
- Nelson DR, Zeldin DC, Hoffman SM, Maltais LJ, Wain HM, Nebert DW (Jan 2004). "Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes and alternative-splice variants". Pharmacogenetics. 14 (1): 1–18. doi:10.1097/00008571-200401000-00001. PMID 15128046. S2CID 18448751.
- Li T, Ma H, Park YJ, Lee YK, Strom S, Moore DD, Chiang JY (Oct 2009). "Forkhead box transcription factor O1 inhibits cholesterol 7alpha-hydroxylase in human hepatocytes and in high fat diet-fed mice". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1791 (10): 991–6. doi:10.1016/j.bbalip.2009.05.004. PMC 2743791. PMID 19463968.
- Chakrabarti B, Dudbridge F, Kent L, Wheelwright S, Hill-Cawthorne G, Allison C, Banerjee-Basu S, Baron-Cohen S (Jun 2009). "Genes related to sex steroids, neural growth, and social-emotional behavior are associated with autistic traits, empathy, and Asperger syndrome". Autism Research. 2 (3): 157–77. doi:10.1002/aur.80. PMID 19598235. S2CID 18239947.
- Barcelos AL, Chies R, Almeida SE, Fiegenbaum M, Schweigert ID, Chula FG, Rossetti ML, Silva CM (Jun 2009). "Association of CYP7A1 -278A>C polymorphism and the response of plasma triglyceride after dietary intervention in dyslipidemic patients". Brazilian Journal of Medical and Biological Research. 42 (6): 487–93. doi:10.1590/s0100-879x2009000600003. PMID 19448895.
- Voora D, Shah SH, Reed CR, Zhai J, Crosslin DR, Messer C, Salisbury BA, Ginsburg GS (Dec 2008). "Pharmacogenetic predictors of statin-mediated low-density lipoprotein cholesterol reduction and dose response". Circulation: Cardiovascular Genetics. 1 (2): 100–6. doi:10.1161/CIRCGENETICS.108.795013. PMC 2995295. PMID 20031551.
- Schwarz M, Lund EG, Russell DW (Apr 1998). "Two 7 alpha-hydroxylase enzymes in bile acid biosynthesis". Current Opinion in Lipidology. 9 (2): 113–8. doi:10.1097/00041433-199804000-00006. PMID 9559267.
- Beigneux A, Hofmann AF, Young SG (Jul 2002). "Human CYP7A1 deficiency: progress and enigmas". The Journal of Clinical Investigation. 110 (1): 29–31. doi:10.1172/JCI16076. PMC 151039. PMID 12093884.
- Ruaño G, Bernene J, Windemuth A, Bower B, Wencker D, Seip RL, Kocherla M, Holford TR, Petit WA, Hanks S (Feb 2009). "Physiogenomic comparison of edema and BMI in patients receiving rosiglitazone or pioglitazone". Clinica Chimica Acta; International Journal of Clinical Chemistry. 400 (1–2): 48–55. doi:10.1016/j.cca.2008.10.009. PMID 18996102.
- Schaap FG, van der Gaag NA, Gouma DJ, Jansen PL (Apr 2009). "High expression of the bile salt-homeostatic hormone fibroblast growth factor 19 in the liver of patients with extrahepatic cholestasis". Hepatology. 49 (4): 1228–35. doi:10.1002/hep.22771. PMID 19185005. S2CID 5306097.
- Srivastava A, Choudhuri G, Mittal B (Jun 2010). "CYP7A1 (-204 A>C; rs3808607 and -469 T>C; rs3824260) promoter polymorphisms and risk of gallbladder cancer in North Indian population". Metabolism. 59 (6): 767–73. doi:10.1016/j.metabol.2009.09.021. PMID 20005541.
External links[edit]
- Cholesterol+7-alpha-Hydroxylase at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P22680 (Cytochrome P450 7A1) at the PDBe-KB.