Coproporphyrinogen III: Difference between revisions
m confirmed chemspider IDs |
Citation bot (talk | contribs) Add: s2cid. | You can use this bot yourself. Report bugs here. | Suggested by Abductive | Category:Tetrapyrroles | via #UCB_Category 18/64 |
||
| Line 42: | Line 42: | ||
In the main [[Porphyrin#Synthesis|porphyrin biosynthesis]] pathway, coproporphyrinogen III is derived from [[uroporphyrinogen III]] by the action of the enzyme [[uroporphyrinogen III decarboxylase]]: |
In the main [[Porphyrin#Synthesis|porphyrin biosynthesis]] pathway, coproporphyrinogen III is derived from [[uroporphyrinogen III]] by the action of the enzyme [[uroporphyrinogen III decarboxylase]]: |
||
[[File:Uroporphyrinogen-decarboxylase.svg|center|480px|alt=Biosynthesis of coproporphyrinogen-III from uroporphyrinogen-III]] |
[[File:Uroporphyrinogen-decarboxylase.svg|center|480px|alt=Biosynthesis of coproporphyrinogen-III from uroporphyrinogen-III]] |
||
The conversion entails four [[decarboxylation]]s, which turn the four acetic acid groups {{chem2|\sCH2\sCOOH}} into methyl groups {{chem2|\sCH3}}, with release of four [[carbon dioxide]] molecules.<ref name=Hemes>{{cite encyclopedia|chapter=Hemes in Biology|author=Paul R. Ortiz de Montellano|year=2008|encyclopedia=Wiley Encyclopedia of Chemical Biology|doi=10.1002/9780470048672.wecb221|publisher=John Wiley & Sons|isbn=978-0470048672}}</ref><ref name=sassa>{{cite journal |pmid=10692079|year=2000|last1=Sassa|first1=S.|title=Molecular aspects of the inherited porphyrias|journal=Journal of Internal Medicine|volume=247|issue=2|pages=169–78|last2=Kappas|first2=A.|doi=10.1046/j.1365-2796.2000.00618.x}}</ref> |
The conversion entails four [[decarboxylation]]s, which turn the four acetic acid groups {{chem2|\sCH2\sCOOH}} into methyl groups {{chem2|\sCH3}}, with release of four [[carbon dioxide]] molecules.<ref name=Hemes>{{cite encyclopedia|chapter=Hemes in Biology|author=Paul R. Ortiz de Montellano|year=2008|encyclopedia=Wiley Encyclopedia of Chemical Biology|doi=10.1002/9780470048672.wecb221|publisher=John Wiley & Sons|isbn=978-0470048672}}</ref><ref name=sassa>{{cite journal |pmid=10692079|year=2000|last1=Sassa|first1=S.|title=Molecular aspects of the inherited porphyrias|journal=Journal of Internal Medicine|volume=247|issue=2|pages=169–78|last2=Kappas|first2=A.|doi=10.1046/j.1365-2796.2000.00618.x|s2cid=36820694}}</ref> |
||
Coproporphyrinogen III is further used as a substrate for the enzyme [[coproporphyrinogen III oxidase]] which oxidizes and further decarboxylates it to [[protoporphyrinogen IX]]. |
Coproporphyrinogen III is further used as a substrate for the enzyme [[coproporphyrinogen III oxidase]] which oxidizes and further decarboxylates it to [[protoporphyrinogen IX]]. |
||
Revision as of 08:42, 12 February 2021
| |
| Names | |
|---|---|
| IUPAC name
3-[8,12,17-tris(2-carboxyethyl)-3,7,13,18-tetramethyl-5,10,15,20,21,22, 23,24-octahydroporphyrin-2-yl]propanoic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| MeSH | Coproporphyrinogen+III |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C36H44N4O8 protonated carboxylic acids | |
| Molar mass | 660.757 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
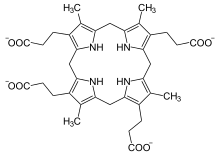
Coproporphyrinogen III is a metabolic intermediate in the biosynthesis of many compounds that are critical for living organisms, such as hemoglobin and chlorophyll. It is a colorless solid.
The compound is a porphyrinogen, a class of compounds characterized by a hexahydroporphine core with various side chains. The coproporphyrinogens have the outermost hydrogen atoms of the core replaced by four methyl groups −CH3 (M) and four propionic acid groups −CH2−CH2−COOH (P). In coproporphyrogen III, the order around the outer ring is MP-MP-MP-PM. For comparison, coproporphyrinogen I has them in the sequence MP-MP-MP-MP. heme.
Biosynthesis and metabolism
In the main porphyrin biosynthesis pathway, coproporphyrinogen III is derived from uroporphyrinogen III by the action of the enzyme uroporphyrinogen III decarboxylase:

The conversion entails four decarboxylations, which turn the four acetic acid groups −CH2−COOH into methyl groups −CH3, with release of four carbon dioxide molecules.[1][2]
Coproporphyrinogen III is further used as a substrate for the enzyme coproporphyrinogen III oxidase which oxidizes and further decarboxylates it to protoporphyrinogen IX.
References
- ^ Paul R. Ortiz de Montellano (2008). "Hemes in Biology". Wiley Encyclopedia of Chemical Biology. John Wiley & Sons. doi:10.1002/9780470048672.wecb221. ISBN 978-0470048672.
- ^ Sassa, S.; Kappas, A. (2000). "Molecular aspects of the inherited porphyrias". Journal of Internal Medicine. 247 (2): 169–78. doi:10.1046/j.1365-2796.2000.00618.x. PMID 10692079. S2CID 36820694.
