Coproporphyrinogen III
| |
| Names | |
|---|---|
| IUPAC name
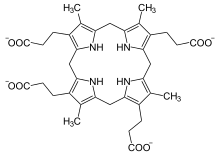
3-[8,12,17-tris(2-carboxyethyl)-3,7,13,18-tetramethyl-5,10,15,20,21,22, 23,24-octahydroporphyrin-2-yl]propanoic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| MeSH | Coproporphyrinogen+III |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C36H44N4O8 | |
| Molar mass | 660.757 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
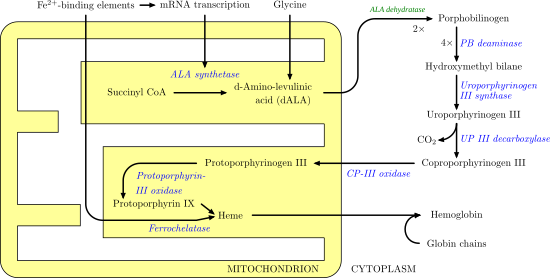
Coproporphyrinogen III in an intermediate in the biosynthesis of natural porphyrins. It is produced from uroporphyrinogen III by the enzyme uroporphyrinogen III decarboxylase. In this process, four carboxymethyl groups (-CH2CO2H) are converted to methyl groups (CH3).
Subsequently coproporphyrinogen III oxidase converts coproporphyrinogen III into protoporphyrinogen IX.[1]
- ^ Paul R. Ortiz de Montellano (2008). "Hemes in Biology". Wiley Encyclopedia of Chemical Biology. John Wiley & Sons. doi:10.1002/9780470048672.wecb221.
