2,5-dibromophenol
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 2,5-dibromophenol | |||||||||||||||
| Molecular formula | C 6 H 4 Br 2 O | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 251.9 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
73-74 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
2,5-Dibromophenol is a chemical compound that belongs to both phenols and halogen aromatic compounds .
presentation
2,5-Dibromophenol can be produced from 2,5-Dibromaniline by diazotization and subsequent treatment with copper sulfate solution.
Derivatives
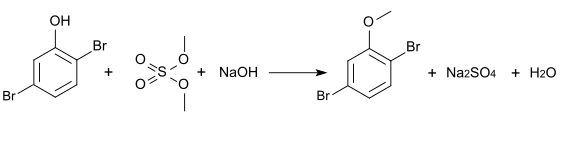
The methyl ether can be produced by methylation with dimethyl sulfate and is also known under the common name 2,5-dibromanisole (CAS number: 95970-08-4).
Individual evidence
- ^ Dictionary of organic compounds, p. 1971 ( limited preview in Google book search).
- ↑ There is not yet a harmonized classification for this substance . A labeling of 2,5-dibromophenol in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on December 3, 2018, is reproduced from a self-classification by the distributor .
- ↑ LO Ruzo, p safe, O. Hutzinger: Metabolism of bromobenzenes in the rabbit. In: J. Agric. Food Chem. , 1976 , 24 (2), pp. 291-293; doi : 10.1021 / jf60204a001 .