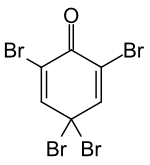
2,4,4,6-tetrabromo-2,5-cyclohexadienone
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 2,4,4,6-tetrabromo-2,5-cyclohexadienone | |||||||||||||||
| other names |
TBCO |
|||||||||||||||
| Molecular formula | C 6 H 2 Br 4 O | |||||||||||||||
| Brief description |
yellow solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 409.70 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| Melting point |
125 ° C (decomposition) |
|||||||||||||||
| solubility |
practically insoluble in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
2,4,4,6-Tetrabromo-2,5-cyclohexadienone is a chemical compound from the group of cyclohexadienones .
Extraction and presentation
2,4,4,6-Tetrabromo-2,5-cyclohexadienone can be obtained by reacting phenol with a brominating agent and a mixture of an alkali / alkaline earth metal bromide and an alkali / alkaline earth metal bromate, dissolved in deionized water in the presence of an acid .
properties
2,4,4,6-Tetrabromo-2,5-cyclohexadienone is a yellow solid that is practically insoluble in water.
use
2,4,4,6-Tetrabromo-2,5-cyclohexadienone has extensive applications in synthetic organic chemistry. It is used in the production of linear poly (phenylene oxides), the direct monobromination of imidazoles and N- methylindoles, the regioselective monobromination of aromatic amines to form 4-bromoanilines in high yields, the parabromination of phenols by the regioselective bromination of phenols for bromination used by thiophenes and many other reactions.
Individual evidence
- ↑ a b c d data sheet 2,4,4,6-Tetrabromo-2,5-cyclohexadienone, 90 +% from AlfaAesar, accessed on March 7, 2018 ( PDF )(JavaScript required) .
- ↑ a b c data sheet 2,4,4,6-tetrabromo-2,5-cyclohexadienone, 90% from Sigma-Aldrich , accessed on March 7, 2018 ( PDF ).
- ↑ a b Document DE60218359T2: METHOD FOR THE PRODUCTION OF 2,4,4,6-TETRABROM-2,5-CYCLOHEXADIENONE , accessed on March 7, 2018.
