2-fluoropropane
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
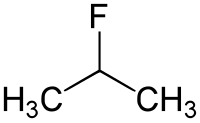
|
||||||||||
| General | ||||||||||
| Surname | 2-fluoropropane | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 3 H 7 F | |||||||||
| Brief description |
colorless gas |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 62.09 g mol −1 | |||||||||
| Physical state |
gaseous |
|||||||||
| Melting point |
−133.4 ° C |
|||||||||
| boiling point |
−10 ° C |
|||||||||
| solubility |
almost insoluble in water |
|||||||||
| Refractive index |
1.3282 |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| Thermodynamic properties | ||||||||||
| ΔH f 0 |
−293.5 kJ / mol |
|||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||
2-fluoropropane is a gaseous organic-chemical compound from the group of fluorocarbons (PFCs).
properties
2-fluoropropane is a flammable gas. It is heavier than air and practically insoluble in water.
literature
- John H. Griffiths, Noel L. Owen, John Sheridan: Rotational and vibrational spectra of 2-fluoropropane. In: J. Chem. Soc., Faraday Trans. 2 69. 1973, pp. 1359-1369; doi : 10.1039 / F29736901359 .
- Michael Meyer, Helmut Dreizler: Internal rotation of molecules with two equivalent tops: The microwave spectrum of 2-fluoropropane, In: Journal of Molecular Spectroscopy 148 (2). 1991, pp. 310-323; doi : 10.1016 / 0022-2852 (91) 90388-Q .
Individual evidence
- ↑ a b c d e data sheet from Apollo Scientific Ltd (PDF; 90 kB), accessed on December 27, 2019.
- ↑ a b Synquestlabs: 2-Fluoropropane - Safety Data Sheet , accessed December 27, 2019.
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Standard Thermodynamic Properties of Chemical Substances, pp. 5-24.

