Chlorobenzotrifluoride
| Chlorobenzotrifluoride | |||||||||
| Surname | 2-chlorobenzotrifluoride | 3-chlorobenzotrifluoride | 4-chlorobenzotrifluoride | ||||||
| other names |
o -chlorobenzotrifluoride 2-chloro-α, α, α-trifluorotoluene o -chloro-α, α, α-trifluorotoluene 1-chloro-2- (trifluoromethyl) benzene |
m -chlorobenzotrifluoride 3-chloro-α, α, α-trifluorotoluene m -chloro-α, α, α-trifluorotoluene 1-chloro-3- (trifluoromethyl) benzene |
p -chlorobenzotrifluoride 4-chloro-α, α, α-trifluorotoluene p -chloro-α, α, α-trifluorotoluene 1-chloro-4- (trifluoromethyl) benzene |
||||||
| Structural formula | 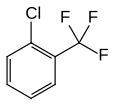 |
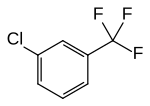 |

|
||||||
| CAS number | 88-16-4 | 98-15-7 | 98-56-6 | ||||||
| ECHA InfoCard | 100.001.641 | 100.002.403 | 100.002.438 | ||||||
| PubChem | 6921 | 7374 | 7394 | ||||||
| Molecular formula | C 7 H 4 ClF 3 | ||||||||
| Molar mass | 180.56 g mol −1 | ||||||||
| Physical state | liquid | ||||||||
| Brief description | yellowish, flammable liquid | colorless, flammable liquid with an aromatic odor | |||||||
| Melting point | −6 ° C | −55.4 ° C | −36 ° C | ||||||
| boiling point | 152 ° C | 137-138 ° C | 139 ° C | ||||||
| density | 1.37 g cm −3 (20 ° C) | 1.336 g cm −3 (25 ° C) | 1.35 g cm −3 (20 ° C) | ||||||
| Vapor pressure | 5 hPa (20 ° C) | 8.5 hPa (20 ° C) | 10.2 hPa (25 ° C) | ||||||
| solubility | practically insoluble in water | ||||||||
|
GHS labeling |
|
|
|
||||||
| H and P phrases | 226-302-315-319-335 | 226-412 | 226-317-411 | ||||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||||
|
210-261-280-301 + 312 302 + 352-305 + 351 + 338 |
no P-phrases | 210-273-280-333 + 313-391-403 + 235 | |||||||
| LD 50 oral rat | > 5,000 mg kg −1 | > 5,000–13,000 mg kg −1 | |||||||
Chlorobenzotrifluorides are chemical compounds from the group of halogenated benzene derivatives .
Extraction and presentation
Chlorobenzotrifluorides can chlorination of benzotrifluoride obtained and subsequent distillation of the isomer mixture. 4-chlorobenzotrifluoride is also obtained by reacting 4-chlorobenzotrichloride and hydrogen fluoride .
use
Chlorobenzotrifluorides are used as intermediate products in the manufacture of pesticides, dyes and pharmaceuticals (e.g. tetrachlorobenzotrifluoride ). So z. B. 4-chlorobenzotrifluoride for the production of dinitroaniline - herbicides such as trifluralin . 4-chlorobenzotrifluoride is also used as a dielectric liquid and as a solvent.
safety instructions
The vapors of chlorobenzotrifluorides can form an explosive mixture with air when heated above their flash point (40–60 ° C).
Derivatives
- 3-nitro-4-chlorobenzotrifluoride
- 3,5-dinitro-4-chlorobenzotrifluoride
- 3-amino-6-chlorobenzotrifluoride
Individual evidence
- ↑ a b c d e f g h Entry for CAS no. 88-16-4 in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d e f g h Entry for CAS no. 98-15-7 in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ a b c d e f g h Entry for CAS no. 98-56-6 in the GESTIS substance database of the IFA , accessed on December 27, 2019(JavaScript required) .
- ↑ Toxicological assessment of m-chlorobenzotrifluoride (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.
- ↑ a b Toxicological assessment of p-chlorobenzotrifluoride (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.


