7-hydroxymitragynine
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
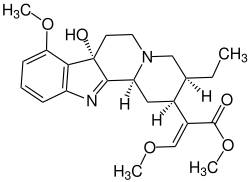
|
|||||||||||||
| General | |||||||||||||
| Surname | 7-hydroxymitragynine | ||||||||||||
| Molecular formula | C 23 H 30 N 2 O 5 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| properties | |||||||||||||
| Molar mass | 414.50 g mol −1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
7-Hydroxymitragynin is in the leaves of the Kratombaumes occurring indole alkaloid and mainly responsible for its psychoactive effects.
Pharmacology and effects
7-Hydroxymitragynine is an opioid analgesic and functions, among other things, as a partial agonist at the μ-opioid receptor. It is about 30 times more potent than mitragynine and 17 times more potent than morphine .
Individual evidence
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Marcus L. Warner, Nellie C. Kaufman, Oliver Grundmann: The pharmacology and toxicology of kratom: from traditional herb to drug of abuse . In: International Journal of Legal Medicine . tape 130 , no. 1 , January 1, 2016, p. 127-138 , doi : 10.1007 / s00414-015-1279-y .
- ↑ Hiromitsu Takayama, Hayato Ishikawa, Mika Kurihara, Mariko Kitajima, Norio Aimi: Studies on the Synthesis and Opioid Agonistic Activities of Mitragynine-Related Indole Alkaloids: Discovery of Opioid Agonists Structurally Different from Other Opioid Ligands . In: Journal of Medicinal Chemistry . tape 45 , no. 9 , April 2002, p. 1949-1956 , doi : 10.1021 / jm010576e .
- ↑ Syunji Horie, Fumi Koyama, Hiromitsu Takayama, Hayato Ishikawa, Norio Aimi: Indole Alkaloids of a Thai Medicinal Herb, Mitragyna speciosa, that has Opioid Agonistic Effect in Guinea-Pig Ileum . In: Planta Medica . tape 71 , no. 3 , March 2005, p. 231-236 , doi : 10.1055 / s-2005-837822 .
