Ammonium hexachlororuthenate
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
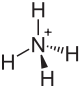 
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Ammonium hexachlororuthenate | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | (NH 4 ) 2 [RuCl 6 ] | |||||||||||||||
| Brief description |
red crystalline powder |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 349.87 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Ammonium hexachlororuthenate is the ammonium salt of hexachlororuthenic acid .
Extraction and presentation
Ammonium hexachlororuthenate (IV) can be obtained by adding a stoichiometric amount of ammonium chloride to a solution obtained by introducing ruthenium (VIII) oxide and chlorine into a dilute hydrogen chloride solution .
properties
(NH 4 ) 2 [RuCl 6 ] forms deep dark red crystals which are sensitive to hydrolysis and which always contain some ammonium pentachlorohydroxoruthenate (IV) (NH 4 ) 2 [RuCl 5 (OH)].
use
Ammonium hexachlororuthenate is an intermediate product in the recycling and recovery of ruthenium . Here, ammonium hexachlororuthenate is obtained from a ruthenium-containing solution and the metal is produced from the sparingly soluble ammonium hexachlororuthenate by calcination and, after several process steps, by reduction with hydrogen .
Individual evidence
- ^ Dale L. Perry, Sidney L. Phillips: Handbook of inorganic compounds .
- ↑ a b data sheet ammonium hexachlororuthenate from Sigma-Aldrich , accessed on June 17, 2017 ( PDF ).
- ↑ a b Georg Brauer (Ed.) U. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume III, Ferdinand Enke, Stuttgart 1981, ISBN 3-432-87823-0 , p. 1749.
- ↑ Patent EP1950178A3: Method for producing ammonium hexachlororuthenate and ruthenium powder, as well as ammonium hexachlororuthenate .
- ↑ Heraeus: technology report 02 (PDF; 566 kB)
