Ammonium nitrite
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
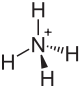 
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Ammonium nitrite | ||||||||||||||||||
| Molecular formula | NH 4 NO 2 | ||||||||||||||||||
| Brief description |
small prisms |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 64.04 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| solubility |
Decomposes in water |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Ammonium nitrite is an ammonium salt of nitrous acid . It has the formula NH 4 NO 2 .
properties
Ammonium nitrite forms colorless, needle-shaped crystals. The connection is unstable and is very easily oxidized by atmospheric oxygen even at room temperature , which can be accelerated even further by high humidity. The crystals turn yellow.
hazards
If the ammonium nitrite is heated in its crystal form, it explodes from a temperature of 60–70 ° C. However, smaller impacts are also sufficient to cause spontaneous explosions.
Reactions
Ammonium nitrite breaks down into nitrogen and water :
use
Ammonium nitrite can be used to synthesize hydroxylamine . It is also suitable as a solid nitrogen source , for example to synthesize salts of NH 2 OH.
Individual evidence
- ↑ a b Entry on ammonium nitrite. In: Römpp Online . Georg Thieme Verlag, accessed on June 27, 2014.
- ↑ There is not yet a harmonized classification for this substance . A labeling of ammonium nitrite in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on July 19, 2019, is reproduced from a self-classification by the distributor .
