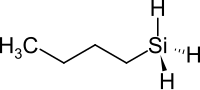
Butylsilane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Butylsilane | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 4 H 12 Si | |||||||||||||||
| Brief description |
colorless liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 88.22 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
0.68 g cm −3 (20 ° C) |
|||||||||||||||
| Melting point |
−138 ° C |
|||||||||||||||
| boiling point |
56.7 ° C |
|||||||||||||||
| Refractive index |
1.392 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Butylsilane is a chemical compound from the group of organosilicon compounds .
Extraction and presentation
Butylsilane can be obtained by reacting monochlorosilane with n -butyllithium ( n BuLi) or by reducing trichlorobutylsilane with lithium aluminum hydride or lithium hydride .
Properties / safety information
Butylsilane is a colorless, flammable liquid with a flash point of −6 ° C that is irritating to the skin, eyes and respiratory tract.
Individual evidence
- ↑ a b c d e f g h Data sheet Butylsilane, ≥97.0% (GC) from Sigma-Aldrich , accessed on October 19, 2015 ( PDF ).
- ↑ a b Barry Arkles: Silanes. (pdf) Reprint from Kirk-Othmer Encyclopedia of Chemical Technology, Forth Edition, Volume 22, pages 38-69. In: Gelest. P. 39 , accessed on December 10, 2016 (English).



