Demelverin
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
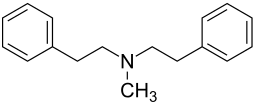
|
||||||||||||||||
| General | ||||||||||||||||
| Non-proprietary name | Demelverin | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 17 H 21 N | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| Drug information | ||||||||||||||||
| Drug class | ||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 239.36 g · mol -1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Demelverine is a drug that is used as an antispasmodic . It shows an effect similar to papaverine . In combination with the anticholinergic Trihexyphenidyl , it was previously marketed under the trade name Spasman ® .
The active ingredient Demelverin was developed by Fahlberg-List in Magdeburg in the 1950s and used until the reunification in the GDR . It was later taken over by Merckle.
application areas
It was used for spastic stomach or intestinal complaints (e.g. irritable bowel syndrome , irritable stomach ), biliary or renal colic and dysmenorrhea . Its application is viewed rather critically nowadays.
Individual evidence
- ↑ There is not yet a harmonized classification for this substance . A labeling of N-methyldiphenethylamine in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), which was accessed on January 15, 2020, is reproduced from a self-classification by the distributor .
- ↑ a b R. Arendt: "Spasman: Rediscovered Spasmolytic". Deutsches Ärzteblatt 94 (1997). Pp. 51-52 ( online ).
- ^ Stiftung Warentest : Spasman solution . 2004. Stiftung Warentest: SPASMAN Scop. Ampullen test.de, December 1, 2012.