Diethylbenzenes
| Diethylbenzenes | ||||||
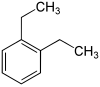
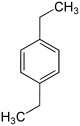
| Surname | 1,2-diethylbenzene | 1,3-diethylbenzene | 1,4-diethylbenzene | |||
| other names | o- diethylbenzene | m- diethylbenzene | p -diethylbenzene | |||
| Structural formula |
|

|
|
|||
| CAS number | 135-01-3 | 141-93-5 | 105-05-5 | |||
| PubChem | 8657 | 8864 | 7734 | |||
| Molecular formula | C 10 H 14 | |||||
| Molar mass | 134.22 g mol −1 | |||||
| Physical state | liquid | |||||
| Melting point | −31.2 ° C | −83.9 ° C | −42.83 ° C | |||
| boiling point | 184 ° C | 181.1 ° C | 183.7 ° C | |||
| solubility | insoluble in water | |||||
|
GHS labeling |
|
|
||||
| H and P phrases | 226 | 226-315-319-335 | ||||
| no EUH phrases | no EUH phrases | |||||
| 210-370 + 378 | 261-305 + 351 + 338 | |||||
In chemistry, diethylbenzenes form a group of substances whose structure consists of a benzene ring with two ethyl groups (–CH 2 –CH 3 ) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 10 H 14 . They also belong to the group of C 4 benzenes .
properties
The boiling points of the diethylbenzenes are almost the same. The melting points differ more clearly. However, 1,4-diethylbenzene, which has the highest symmetry, does not have the highest melting point.
Web links
Commons : Diethylbenzenes - Collection of pictures, videos and audio files
Individual evidence
- ↑ a b c d e f David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Physical Constants of Organic Compounds, pp. 3-166.
- ↑ Data sheet 1,2-Diethylbenzene from Sigma-Aldrich , accessed on May 15, 2017 ( PDF ).
- ↑ Data sheet 1,3-Diethylbenzene from Sigma-Aldrich , accessed on May 15, 2017 ( PDF ).
- ↑ Data sheet 1,4-Diethylbenzene from Sigma-Aldrich , accessed on May 15, 2017 ( PDF ).

