Dihydroxytoluenes
The dihydroxytoluenes form a group of aromatic compounds that are derived from both toluene and dihydroxybenzenes . The structure consists of a benzene ring with two attached hydroxyl groups (-OH) and a methyl group (-CH 3 ) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 7 H 8 O 2 , the best known being 3,5-dihydroxytoluene ( orcin ).
A uniform naming is difficult because the dihydroxytoluenes do not form a common common name. They can therefore be understood as derivatives of catechol (2 isomers), resorcinol (3 isomers) and hydroquinone (1 isomer). Conversely, they can be viewed as dihydroxy derivatives of toluene. They are therefore referred to as dihydroxytoluenes , since only -toluene forms a common new common tribe name .
Occurrence
The 3,4- isomer homopyrocatechin (also homobrenzcatechin, 4-methylpyrocatechin) is found as 2-methylether creosol , on the one hand in creosote , in wood tar and in ylang-ylang oil . The 3,5-isomer orcin was isolated in 1829 from lichens of the genus Rocella . It is also found in the flowers of mosses such as Ditrichia viscosa and the leaves of tree heather ( Erica arborea ).
properties
All substances are sparingly soluble in water. Chemically, they typically behave like phenols, for example in alkaline solution they form phenolates, which have a significantly higher water solubility.
| Dihydroxytoluenes | ||||||||||||
| Surname | 2,3-dihydroxytoluene | 2,4-dihydroxytoluene | 2,5-dihydroxytoluene | 2,6-dihydroxytoluene | 3,4-dihydroxytoluene | 3,5-dihydroxytoluene | ||||||
| other names | 3-methyl- 1,2-dihydroxybenzene, 3-methylpyrocatechol |
4-methyl- 1,3-dihydroxybenzene, 4-methylresorcinol |
2-methyl- 1,4-dihydroxybenzene, 2-methylhydroquinone, toluhydroquinone |
2-methyl- 1,3-dihydroxybenzene, 2-methylresorcinol |
4-methyl- 1,2-dihydroxybenzene, 4-methylpyrocatechol, homopyrocatechol |
5-methyl- 1,3-dihydroxybenzene, 5-methylresorcinol, orcin , orcinol |
||||||
| Structural formula |

|

|
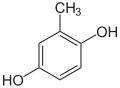
|

|
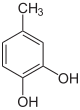
|

|
||||||
| CAS number | 488-17-5 | 496-73-1 | 95-71-6 | 608-25-3 | 452-86-8 | 504-15-4 | ||||||
| PubChem | 340 | 10333 | 7253 | 11843 | 9958 | 10436 | ||||||
| ECHA InfoCard | 100.006.975 | 100.007.117 | 100.002.222 | 100.009.233 | 100.006.559 | 100.007.259 | ||||||
| Molecular formula | C 7 H 8 O 2 | |||||||||||
| Molar mass | 124.14 g mol −1 | |||||||||||
| Physical state | firmly | |||||||||||
| Brief description | crystalline solids | |||||||||||
| Melting point | 65-68 ° C | 104-108 ° C | 126-127 ° C | 114-120 ° C | 67-69 ° C | 56-58 ° C | ||||||
| boiling point | 241 ° C | 270 ° C | 297 ° C | 264 ° C | 251 ° C | 290 ° C | ||||||
| solubility | slightly soluble in water | |||||||||||
|
GHS labeling |
|
|
|
|
|
|
||||||
| H and P phrases | 315-319-335 | 315-319-335 | 302-315-319-335 | 301-315-319-335 | 315-319-335 | 302-315-319-335 | ||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | |||||||
| 261-305 + 351 + 338 | 261-305 + 351 + 338 | 261-305 + 351 + 338 | 261-301 + 310-305 + 351 + 338 | 261-305 + 351 + 338 | 261-305 + 351 + 338 | |||||||
Individual evidence
- ↑ H. Hager et al .: Hagers Handbook of Pharmaceutical Practice , Springer 1999, ISBN 3-540-52640-4 , p. 681.
- ↑ J. Falbe, M. Regitz (ed.): Römpp Lexikon Chemie , 10th edition, Thieme, Stuttgart a. New York 1996-1999, p. 2642.
- ↑ a b c data sheet 3-methylcatechol from Sigma-Aldrich , accessed on December 26, 2019 ( PDF ).
- ↑ a b Data sheet 4-methylresorcinol from Sigma-Aldrich , accessed on December 27, 2019 ( PDF ).
- ↑ a b c data sheet methylhydroquinone from Sigma-Aldrich , accessed on December 26, 2019 ( PDF ).
- ↑ a b c data sheet 2-methylresorcinol from Sigma-Aldrich , accessed on December 26, 2019 ( PDF ).
- ↑ a b c data sheet 4-methylcatechol from Sigma-Aldrich , accessed on December 26, 2019 ( PDF ).
- ↑ a b c data sheet Orcinol monohydrate from Sigma-Aldrich , accessed on December 26, 2019 ( PDF ).
- ↑ David R. Lide: CRC Handbook of Chemistry and Physics A Ready-reference Book of Chemical and Physical Data . CRC Press, 1995, ISBN 978-0-8493-0595-5 , pp. 370 ( limited preview in Google Book search).

