Dropropizine
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
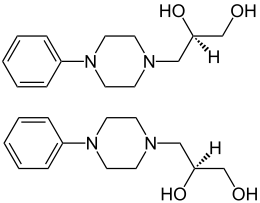
|
||||||||||||||||
| 1: 1 mixture of ( R ) -form (top) and ( S ) -form (bottom) | ||||||||||||||||
| General | ||||||||||||||||
| Non-proprietary name | Dropropizine | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 13 H 20 N 2 O 2 | |||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| Drug information | ||||||||||||||||
| ATC code | ||||||||||||||||
| Drug class | ||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 236.31 g · mol -1 | |||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Dropropizin (or Dipropizin) (trade name: Larylin Husten-Stiller ® ) is an active ingredient from the group of antitussives ( antitussive agents). It is used for irritable and convulsive coughs and irritations of the upper respiratory tract . The active ingredient must not be used in the case of impaired liver or kidney function, severe cardiovascular problems and children under 12 years of age. There is no experience on use during pregnancy or breastfeeding. At high doses, there may be a short-term drop in blood pressure and a racing heart . Occasionally, tiredness and mild diarrhea may occur, rarely allergic reactions .
Dropropizin contains a stereocenter on the propanediol residue . It is used as a racemate [1: 1 mixture of the ( R ) enantiomer and the ( S ) enantiomer]. The effective ( S ) -enantiomer ( eutomer ) is levodropropizine and is also used as a cough suppressant. The market launch of levodropropizin in 2000 was justified with a reduction in side effects with the same dosage. To achieve a comparable therapeutic effect, about half the dose of levodropropizine is sufficient compared to dropropizine.
The mechanism of action is not yet fully understood.
Individual evidence
- ↑ a b data sheet (±) -Dropropizine from Sigma-Aldrich , accessed on November 7, 2016 ( PDF ).
- ↑ Rote Liste Service GmbH (Ed.): Rote Liste 2017 - drug directory for Germany (including EU approvals and certain medical devices) , Rote Liste Service GmbH, Frankfurt / Main, 2017, edition 57, ISBN 978-3-946057-10 -9 , p. 178.