Enoxastrobin
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
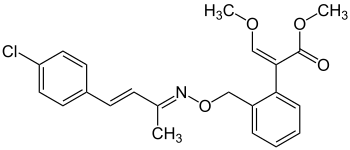
|
||||||||||
| General | ||||||||||
| Surname | Enoxastrobin | |||||||||
| other names |
|
|||||||||
| Molecular formula | C 22 H 22 ClNO 4 | |||||||||
| Brief description |
white solid |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 399.87 g mol −1 | |||||||||
| Physical state |
firmly |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Enoxastrobin is a chemical compound from the group of methoxyacrylates and strobilurins .
use
Enoxastrobin is used as a fungicide . It works by inhibiting cell respiration (QoL fungicide). The compound was discovered by the Rohm and Haas Company and examined by the Shenyang Research Institute of Chemical Industry.
In Germany, Austria and Switzerland, no pesticides with this active ingredient are permitted.
Individual evidence
- ↑ Ulrich Schirmer, Peter Jeschke, Matthias Witschel: Modern Crop Protection Compounds: Herbicides . John Wiley & Sons, 2012, ISBN 978-3-527-32965-6 , pp. 607 ( limited preview in Google Book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ↑ Entry on enoxastrobin in the Pesticide Properties DataBase (PPDB) of the University of Hertfordshire , accessed on February 13, 2015.
- ↑ Song Tu, Ya-Qiang Xie, Si-Zhe Gui, Li-Yi Ye, Zi-Long Huang, Yi-Bing Huang, Li-Ming Che: Synthesis and fungicidal activities of novel benzothiophene-substituted oxime ether strobilurins. In: Bioorganic & Medicinal Chemistry Letters. 24, 2014, pp. 2173-2176, doi : 10.1016 / j.bmcl.2014.03.024 .
- ^ Directorate-General for Health and Food Safety of the European Commission: EU pesticide database ; Entry in the national registers of plant protection products in Switzerland , Austria and Germany ; accessed on February 14, 2016.