Goserelin
| Goserelin | ||
|---|---|---|
|
||
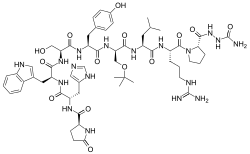
| Structural formula | ||
| Mass / length primary structure | 10 amino acids | |
| Identifier | ||
| External IDs |
|
|
| Drug information | ||
| ATC code | L02 AE03 | |
| DrugBank | DB00014 | |
| Drug class | Antiandrogens | |
Goserelin (trade name Zoladex ® ; manufacturer AstraZeneca ) is a drug that is used to suppress sex hormones such as estrogen and testosterone . It belongs to the group of analogs of the gonadotropin-releasing hormone , which is used, among other things, in the treatment of prostate and breast cancer .
Clinical information
Application areas (indications)
Areas of application for goserelin are prostate cancer, breast cancer, transsexuality , endometriosis (mostly benign growth of the uterine lining ), uterine myoma (benign tumor of the uterus) and endometrial ablation (removal of the lining of the uterus). The active ingredient is also used in reproductive medicine.
Use during pregnancy and breastfeeding
Goserelin should not be used during pregnancy, as there is theoretically a risk of miscarriage or deformity of the unborn child. Goserelin should also not be used during breastfeeding .
Adverse effects (side effects)
- Decrease in bone density
- depression
- Decrease in glucose tolerance
- high blood pressure
Pharmacological properties
Mechanism of action (pharmacodynamics)
Goserelin overstimulates the pituitary gland , so that initially more luteinizing hormone and follicle-stimulating hormone and thus estrogens are formed, whereby testosterone is broken down into estrogen. However, the hormone production subsides after about 2–4 weeks.
Trade names
- Monopreparations
- Zoladex (D, A, CH)
Individual evidence
- ↑ Björn Lemmer , Georges Fülgraff (Ed.): Pharmacotherapy, clinical pharmacology . 14., revised. and updated edition. Springer, Heidelberg 2010, ISBN 978-3-642-10540-1 , pp. 165 .