Hafnium (IV) iodide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
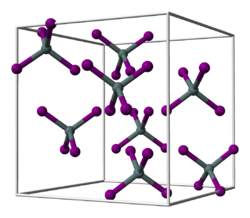
|
||||||||||||||||
| __ Hf 4+ __ I - | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Hafnium (IV) iodide | |||||||||||||||
| other names |
Hafnium tetraiodide |
|||||||||||||||
| Ratio formula | HfI 4 | |||||||||||||||
| Brief description |
yellow-orange solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 686.11 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
5.6 g cm −3 (25 ° C) |
|||||||||||||||
| Melting point |
455 ° C (under pressure) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Hafnium (IV) iodide is an inorganic chemical compound of hafnium from the group of iodides .
Extraction and presentation
Hafnium (IV) iodide can be produced by reacting hafnium with iodine
or obtained by reacting hafnium (IV) chloride with aluminum iodide.
properties
Hafnium (IV) iodide is a yellow-orange solid that hydrolyzes in water to form hafnium oxide iodide and has a crystal structure of the tin (IV) iodide type with the space group Pa 3 (space group no. 205) . It sublimes at 393 ° C.
use
Hafnium (IV) iodide is used to make pure hafnium. In 1925 the first sample of pure hafnium was obtained by pyrolysis of hafnium (IV) iodide.
Individual evidence
- ↑ a b c d e Georg Brauer (Hrsg.): Handbook of Preparative Inorganic Chemistry . 3., reworked. Edition. tape II . Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 1360 .
- ↑ a b c d Data sheet Hafnium (IV) iodide, anhydrous, 99% from Sigma-Aldrich , accessed on May 4, 2013 ( PDF ).
- ^ Alan George Sharpe: Inorganic chemistry. [Main volume] Pearson Education, 2005, ISBN 978-0-13-039913-7 , pp. 652 ( limited preview in Google Book search).
- ↑ Roger Blachnik (Ed.): Paperback for chemists and physicists . Volume III: Elements, Inorganic Compounds and Materials, Minerals . founded by Jean d'Ans, Ellen Lax. 4th, revised and revised edition. Springer, Berlin 1998, ISBN 3-540-60035-3 , pp. 478 ( limited preview in Google Book search).
- ^ Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis US, 2011, ISBN 1-4398-1462-7 , pp. 194 ( limited preview in Google Book search).
- ^ Mark T. Swihart, Mark Donald Allendorf, M. Meyyappan: Fundamental Gas-Phase and Surface Chemistry of Vapor-Phase Deposition II and ... The Electrochemical Society, 2001, ISBN 978-1-56677-319-5 , pp. 153 ( limited preview in Google Book search).
- ↑ Z. Guide to the Elements: Nature's Building Blocks: An A . Oxford University Press, 2001, ISBN 0-19-850341-5 , pp. 173 ( limited preview in Google Book search).


