Hexanols
In chemistry, the hexanols form a group of aliphatic , saturated , simple alcohols ( alkanols ) with 6 carbon atoms and thus the empirical formula C 6 H 13 OH. There are 17 structural isomers . The isomer mixture has the CAS number 25917-35-5.
Structural formula IUPAC name Type Alkane framework Melting point (° C) Boiling point (° C) 
Hexan-1-ol Primary n -hexane −45 157 
Hexan-2-ol Secondary n -hexane −50 140 Hexan-3-ol Secondary n -hexane 6th 135 
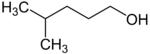
2-methylpentan-1-ol Primary 2-methylpentane −107 148 3-methylpentan-1-ol Primary 3-methylpentane 152 4-methylpentan-1-ol Primary 2-methylpentane 151 2-methylpentan-2-ol Tertiary 2-methylpentane 121 3-methylpentan-2-ol Secondary 3-methylpentane 134 4-methylpentan-2-ol Secondary 2-methylpentane 131 2-methylpentan-3-ol Secondary 2-methylpentane 126 
3-methylpentan-3-ol Tertiary 3-methylpentane −24 122 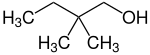
2,2-dimethylbutan-1-ol Primary 2,2-dimethylbutane −22 137 2,3-dimethylbutan-1-ol Primary 2,3-dimethylbutane 145 3,3-dimethylbutan-1-ol Primary 2,2-dimethylbutane −39 143 2,3-dimethylbutan-2-ol Tertiary 2,3-dimethylbutane 119 3,3-dimethylbutan-2-ol Secondary 2,2-dimethylbutane 120 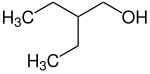
2-ethylbutan-1-ol Primary 3-methylpentane 146