Iodobenzoic acids
| Iodobenzoic acids | |||||||
| Surname | 2-iodobenzoic acid | 3-iodobenzoic acid | 4-iodobenzoic acid | ||||
| other names | o -iodobenzoic acid | m -iodobenzoic acid | p -iodobenzoic acid | ||||
| Structural formula |

|
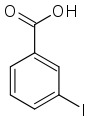
|
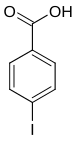
|
||||
| CAS number | 88-67-5 | 618-51-9 | 619-58-9 | ||||
| PubChem | 6941 | 12060 | 12085 | ||||
| Molecular formula | C 7 H 5 IO 2 | ||||||
| Molar mass | 248.02 g mol −1 | ||||||
| Physical state | firmly | ||||||
| Brief description | colorless crystals | ||||||
| Melting point | 160-162 ° C | 185-187 ° C | 270-273 ° C | ||||
| pK s value | 2.86 | 3.85 | - | ||||
|
GHS labeling |
|
|
|
||||
| H and P phrases | 302-315-318-335 | no H-phrases | 315-332-335 | ||||
| no EUH phrases | no EUH phrases | no EUH phrases | |||||
| 261-280-305 + 351 + 338 | no P-phrases | 261-305 + 351 + 338 | |||||
In chemistry , the iodobenzoic acids form a group of substances that are derived from both benzoic acid and iodobenzene . The structure consists of a benzene ring with an attached carboxy group (–COOH) and iodine (–I) as substituents . Their different arrangement results in three constitutional isomers with the empirical formula C 7 H 5 IO 2 .
presentation
The iodobenzoic acids can be prepared from the corresponding aminobenzoic acids by diazotization and subsequent addition of iodide solutions ( Sandmeyer reaction ).
properties
The iodobenzoic acids are crystalline solids. The melting points differ significantly. The 4-iodobenzoic acid, which has the highest symmetry, has the highest melting point. The iodobenzoic acids have a higher acidity than benzoic acid due to the −I effect of the iodine substituent. The pK s values are therefore correspondingly lower (benzoic acid: 4.20).
Individual evidence
- ↑ a b Harold P. Klug: "The Crystal Structure of Ortho-Iodobenzoic Acid", in: J. Am. Chem. Soc. , 1933 , 55 (4), pp. 1430-1436; doi: 10.1021 / ja01331a015 .
- ↑ a b Data sheet 2-Iodobenzoic acid from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b Data sheet 3-Iodobenzoic acid from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b Data sheet 4-Iodobenzoic acid from Sigma-Aldrich , accessed on May 3, 2011 ( PDF ).
- ↑ a b CRC Handbook of Tables for Organic Compound Identification , Third Edition, 1984, ISBN 0-8493-0303-6 .
- ↑ Hubertus Peter Bell: Palladium-catalyzed domino cyclizations for the efficient total synthesis of tetracycline antibiotics . Cuvillier Verlag, Göttingen 2004, ISBN 978-3-86537-079-2 , p. 45 ( limited preview in Google book search - plus dissertation, University of Göttingen).
- ↑ Harold P. Klug, Edward Mack Jr., FC Blake: "The Crystal Structure of Meta-Iodobenzoic Acid", in: J. Am. Chem. Soc. , 1929 , 51 (10), pp. 2880-2888; doi: 10.1021 / ja01385a003 .
- ↑ Russell G. Baughman, Janet E. Nelson: “The structure of p- iodobenzoic acid, C 7 H 5 IO 2 ”, in: Acta Crystallographica C , 1984 , 40 , pp 204-206; doi: 10.1107 / S0108270184003607 .

