Isononanoic acid
Isononanoic acid is the name for the technical mixture of the branched structural isomers of nonanoic acid , a C 9 carboxylic acid.
Extraction and presentation
Technical isononanoic acid is produced by the oxidation of isononanaldehydes , which are produced from diisobutene . An isomer mixture is formed, which consists of more than 90% 3,5,5-trimethylhexanoic acid . If, on the other hand, diisobutene is reacted directly with carbon monoxide in a Koch-Haaf synthesis (85% sulfuric acid , 20 ° C., 7 bar ), 2,2,4,4-tetramethylpentanoic acid is mainly formed . There is also 7-methyloctanoic acid, the actual isononanoic acid.
Structural isomers
| Structural isomers | ||||
| Surname | 3,5,5-trimethylhexanoic acid | 2,2,4,4-tetramethylpentanoic acid | 7-methyloctanoic acid | |
| Structural formula |

|

|
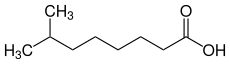
|
|
| CAS number | 3302-10-1 | 3302-12-3 | 693-19-6 | |
| 26896-18-4 (mixture) | ||||
| Molecular formula | C 9 H 18 O 2 | |||
| Molar mass | 158.24 g mol −1 | |||
| EC number | 221-975-0 | |||
| 248-092-3 (mixture) | ||||
| ECHA info card | 100,019,978 | |||
| 100.043.706 (mixture) | ||||
| PubChem | 90960 | 18699 | 33635 | |
| Wikidata | Q15725607 | Q53864639 | Q27117040 | |
| Q53864465 (mixture) | ||||
Physical Properties
The physical properties of technical isononanoic acid vary depending on the composition. The melting point is between -60 ° C and -70 ° C, the boiling point between 228 ° C and 255 ° C, the density between 0.895 and 0.902 g / cm 3 and the vapor pressure between 0.1 and 0.01 hPa .
use
Isononanoic acid is used in the manufacture of alkyd resins and siccatives for varnishes and oil paints. It is an intermediate stage in the production of isononanyl peroxide and plays a role in the production of PVC polymers. It is also a precursor for fungicides and a cosmetic base.
Individual evidence
- ↑ a b Entry on isononanoic acid. In: Römpp Online . Georg Thieme Verlag, accessed on May 20, 2018.
- ↑ a b c Toxicological assessment of isononanoic acid (PDF) at the professional association for raw materials and chemical industry (BG RCI), accessed on May 20, 2018.