Pyrosulfuryl chloride
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
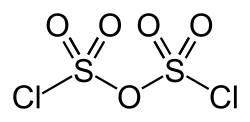
|
||||||||||
| General | ||||||||||
| Surname | Pyrosulfuryl chloride | |||||||||
| other names |
Disulfuryl chloride |
|||||||||
| Molecular formula | Cl 2 O 5 S 2 | |||||||||
| Brief description |
colorless liquid with a peculiar smell |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 215.03 g mol −1 | |||||||||
| Physical state |
liquid |
|||||||||
| density |
1.84 g cm −3 |
|||||||||
| Melting point |
−37 ° C |
|||||||||
| boiling point |
152-153 ° C |
|||||||||
| Refractive index |
1,837 |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||
Pyrosulfuryl chloride is a chemical compound from the group of acid chlorides . It is the anhydride of chlorosulfonic acid .
Extraction and presentation
Pyrosulfuryl chloride can be obtained by reacting sulfur trioxide with carbon tetrachloride .
properties
Pyrosulfuryl chloride is a colorless, easily mobile liquid with a characteristic odor, which faintly smokes in moist air and becomes cloudy with the separation of sulfuric acid . With prolonged boiling or heating to 250 ° C, dissociation occurs into sulfur trioxide, sulfur dioxide and chlorine .
Individual evidence
- ^ A b Jean d'Ans, Ellen Lax, Roger Blachnik: Pocket book for chemists and physicists . Springer DE, 1998, ISBN 3-642-58842-5 , pp. 710 ( limited preview in Google Book search).
- ↑ a b c d e f Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 389.
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
